Question: (Postacidification in yogurt) Yogurt is produced by adding two strains of bacteria (Lactobacillus bulgaricus and Streptococcus thermophilus) to pasteurized milk. At temperatures of 11O
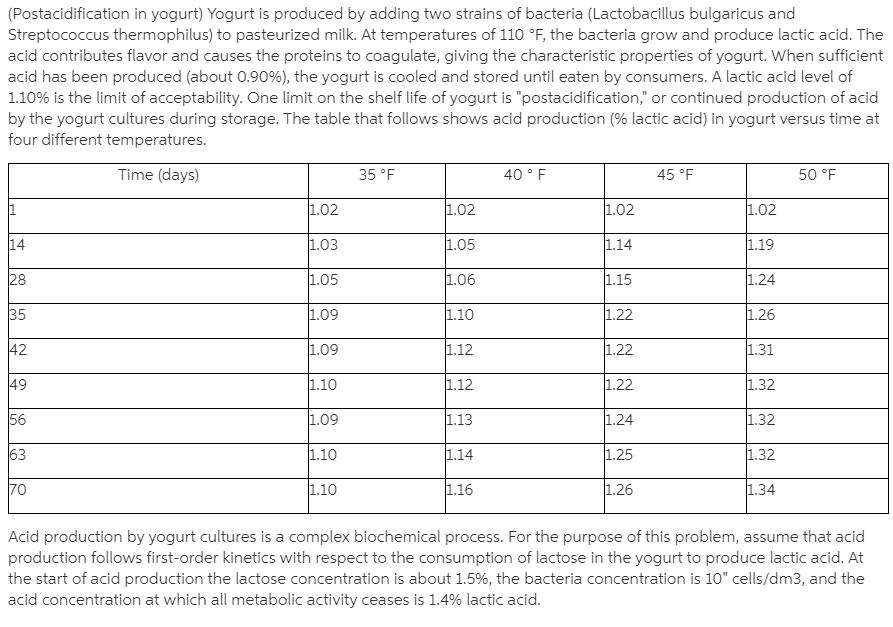

(Postacidification in yogurt) Yogurt is produced by adding two strains of bacteria (Lactobacillus bulgaricus and Streptococcus thermophilus) to pasteurized milk. At temperatures of 11O F, the bacteria grow and produce lactic acid. The acid contributes flavor and causes the proteins to coagulate, giving the characteristic properties of yogurt. When sufficient acid has been produced (about 0.90%), the yogurt is cooled and stored until eaten by consumers. A lactic acid level of 1.10% is the limit of acceptability. One limit on the shelf life of yogurt is "postacidification," or continued production of acid by the yogurt cultures during storage. The table that follows shows acid production (% lactic acid) in yogurt versus time at four different temperatures. 40 F Time (days) 35 F 45 F 50 F 1.02 1.02 1.02 1.02 1.05 14 1.03 1.14 1.19 28 1.15 1.05 1.06 1.24 1.09 1.10 1.22 1.26 35 1.12 1.22 42 1.31 1.09 49 1.10 1.32 1.12 1.22 56 1.09 1.13 1.24 1.32 63 1.10 1.14 1.25 1.32 1.16 1.26 70 1.10 1.34 Acid production by yogurt cultures is a complex biochemical process. For the purpose of this problem, assume that acid production follows first-order kinetics with respect to the consumption of lactose in the yogurt to produce lactic acid. At the start of acid production the lactose concentration is about 1.5%, the bacteria concentration is 10" cells/dm3, and the acid concentration at which all metabolic activity ceases is 1.4% lactic acid. (a) Determine the activation energy for the reaction. (b) How long would it take to reach 1.10% acid at 38 F? (c) If you left yogurt out at room temperature, 77 F, how long would it take to reach 1.10% lactic acid? (d) Assuming that the lactic acid is produced in the stationary state, do the data fit any of the modules developed in this chapter? [Problem developed by General Mills, Minneapolis, Minnesota] (e) List ways you can work this problem incorrectly. (f) How could you make this problem more difficult?
Step by Step Solution
3.53 Rating (150 Votes )
There are 3 Steps involved in it
To solve the problem lets tackle each part step by step a Determine the activation energy for the reaction To determine the activation energy Ea we ca... View full answer

Get step-by-step solutions from verified subject matter experts


