The most abundant radon isotope is radon-222, which is an alpha emitter with a half-life of...
Fantastic news! We've Found the answer you've been seeking!
Question:

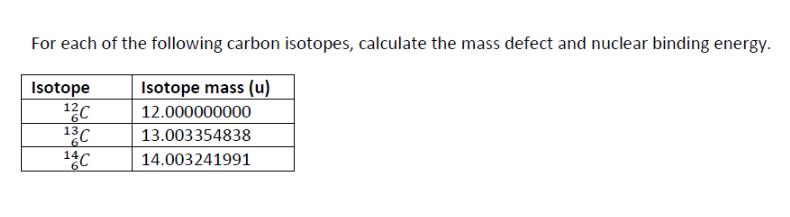
Transcribed Image Text:
The most abundant radon isotope is radon-222, which is an alpha emitter with a half-life of 3.8 days. The air in a house basement in Canada is found to have a radon level of 500 Bq m³. The World Health Organization's safe limit for radon is 100 Bq m³. How long will it take for the radon level to decay to 100 Bq m³ in a sample of air from this basement? An archaeologist uncovers a wooden object at an archaeological site. The object is found to have a carbon-14 decay rate of 0.11 disintegrations per second per gram of carbon. How old is the object? (Living organisms have a decay rate of 0.255 s¹ g¹, and t₁/2 = 5730 y for C-14.) Thorium-232 has a radioactive decay series that ultimately produces lead-208. What is the expected ratio of 202Pb/232Th in a meteorite that is approximately 3.0 × 109 years old? (The half-life of thorium-232 is 1.4 x 1010 y.) For each of the following carbon isotopes, calculate the mass defect and nuclear binding energy. Isotope 12C Isotope mass (u) 12.000000000 13C 13.003354838 6 14C 14.003241991 The most abundant radon isotope is radon-222, which is an alpha emitter with a half-life of 3.8 days. The air in a house basement in Canada is found to have a radon level of 500 Bq m³. The World Health Organization's safe limit for radon is 100 Bq m³. How long will it take for the radon level to decay to 100 Bq m³ in a sample of air from this basement? An archaeologist uncovers a wooden object at an archaeological site. The object is found to have a carbon-14 decay rate of 0.11 disintegrations per second per gram of carbon. How old is the object? (Living organisms have a decay rate of 0.255 s¹ g¹, and t₁/2 = 5730 y for C-14.) Thorium-232 has a radioactive decay series that ultimately produces lead-208. What is the expected ratio of 202Pb/232Th in a meteorite that is approximately 3.0 × 109 years old? (The half-life of thorium-232 is 1.4 x 1010 y.) For each of the following carbon isotopes, calculate the mass defect and nuclear binding energy. Isotope 12C Isotope mass (u) 12.000000000 13C 13.003354838 6 14C 14.003241991
Expert Answer:

Related Book For 

Auditing a risk based approach to conducting a quality audit
ISBN: 978-1133939153
9th edition
Authors: Karla Johnstone, Audrey Gramling, Larry Rittenberg
Posted Date:
Students also viewed these finance questions
-
The Crazy Eddie fraud may appear smaller and gentler than the massive billion-dollar frauds exposed in recent times, such as Bernie Madoffs Ponzi scheme, frauds in the subprime mortgage market, the...
-
Do firms follow the same steps for impairment testing of finite- and indefinite- life intangible assets ? Explain.
-
A torque of 0.12 N m is applied to an egg beater. (a) If the egg beater starts at rest, what is its angular momentum after 0.65 s? (b) If the moment of inertia of the egg beater is 2.5 10-3 kg m2,...
-
Bob sells a stock investment for $35,000 cash, and the purchaser assumes Bobs $32,500 debt on the investment. The basis of Bobs stock investment is $55,000. What is the gain or loss realized on the...
-
Pick an industry and a product or service. Engage in a creative-thinking process, as outlined in Chapter 11, to generate an improved offering. Do the same to create an entirely new offering that uses...
-
Alternative methods of joint-cost allocation, product-mix decisions. The Southern Oil Company buys crude vegetable oil. Refining this oil results in four products at the splitoff point: A, B, C, and...
-
Calculate the dividends paid per share of common stock. ( Note: Number of shares shown on balance sheet is not shown in thousands. The number of shares " in thousands" is 2 , 0 0 0 . ) Round...
-
A one-dimensional harmonic oscillator has an infinite series of equally spaced energy states, with E x = sh, where s is a positive integer or zero, and is the classical frequency of the oscillator....
-
Suppose a utility generates electricity with a 32% efficient coal-fired power plant that emits SO at the legal limit of 0.6 lb per million Btu of heat into the plant. If the utility customers replace...
-
What is lactose intolerance? Also why is it, emically spea 7) When lactose and water react, what are the products? Where in the digestive system does this reaction 8) What is the group of disorders...
-
Chrome File Edit Arial Name Partner (if applicable) Bookmarks Profiles Tab dit Vi View History Chem 111 Experiment 7 Online Lab Report.docx esc Volume of vinegar solution (mL) Mass of vinegar...
-
What will the volume of 111 mol of nitrogen in the stratosphere, where the temperature is -57C and the pressure is 7.30 kPa? 4. How many moles of a gas are in a balloon that has a volume of 15.9 L at...
-
In organic chemistry reaction mechanisms, how do you know when to have the oxygen on the carobnyl group attack a hydrogen as compared to using its resonance structure ( going to a single bond ) ? Ht...
-
If a saturated solution of sodium nitrate, NaNO3, is prepared, the following equilibrium exists: a) NaNO3 (s) Na+ (aq) + NO3(aq) = If nitric acid is added to the saturated solution, what will happen...
-
Relations Rand s are defined by R = ((1,5), (2,2), (3,4), (5,2)} and s = ((2,4), (3,4), (3,1), (5,5)). Then Dom(s o R) : %3D %3D %3D 0 (2,3,5) (1,2,3) o (1,2,5) O (1,2,3,4) QUESTION 5 Let be the...
-
Per Bag Direct materials: 25 pounds of CWhiz-2000 @ $0.08/lb. = $ 2.00 Direct labor: 0.05 hour @ $32.00/hr. = $ 1.60 The company manufactured 100,000 bags of Cheese-Be-Good in December and used...
-
Match the following assertions with their associated description: (a) Existence or occurrence, (b) Completeness, (c) Rights and obligations, (d) Valuation or allocation, (e) Presentation and...
-
On February 28, 2014, Stu & Dent, LLP completed the audit of Shylo Ranch, Inc. (a public company) for the year ended December 31, 2013. A recent fire destroyed the accounting records concerning the...
-
Why is it important to assess whether potential misclassifications in the statement of cash flows are material?
-
Select an information system with which you are familiar, and which you feel needs to be improved, based upon your experiences as an employee, customer, other system user, or system owner, Switch...
-
You have learned the importance of making sure that requirements are correctly identified. Hut how do you know when you have a correct requirement-that is, what criteria must each requirement meet in...
-
Prepare a full feasibility analysis, including Economic, Operational, Schedule, Legal, and Technical analyses for the system you are suggesting in problem 1. Your analysis should be no more than 30...

Study smarter with the SolutionInn App


