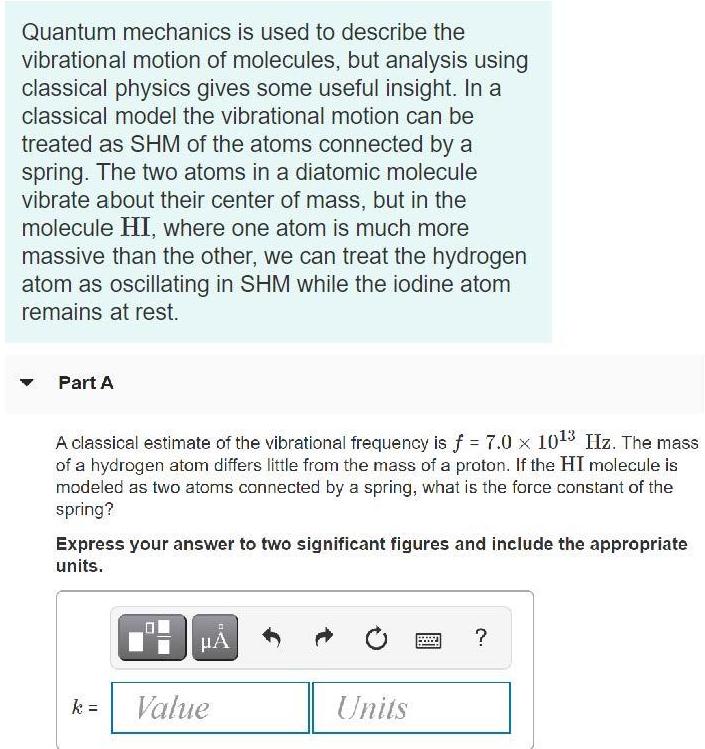
Quantum mechanics is used to describe the vibrational motion of molecules, but analysis using classical physics...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
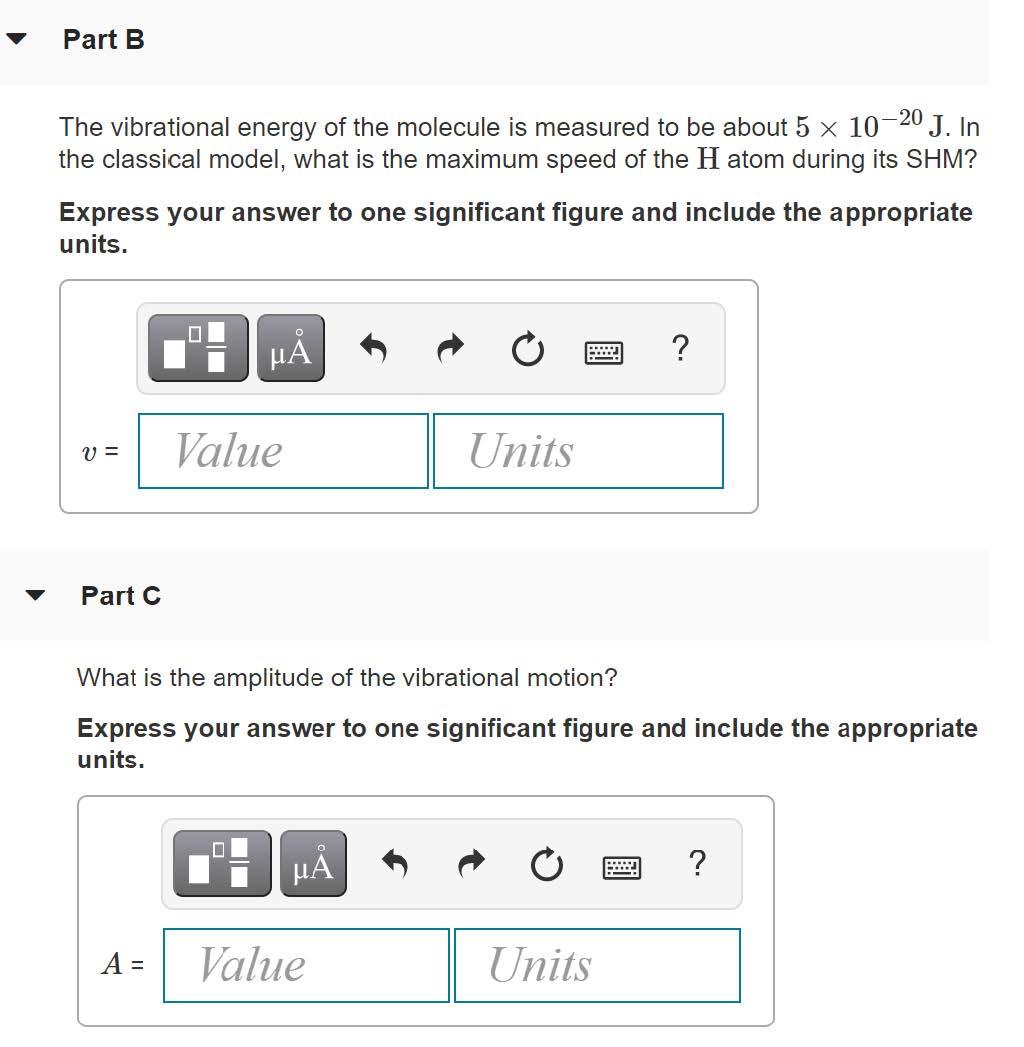
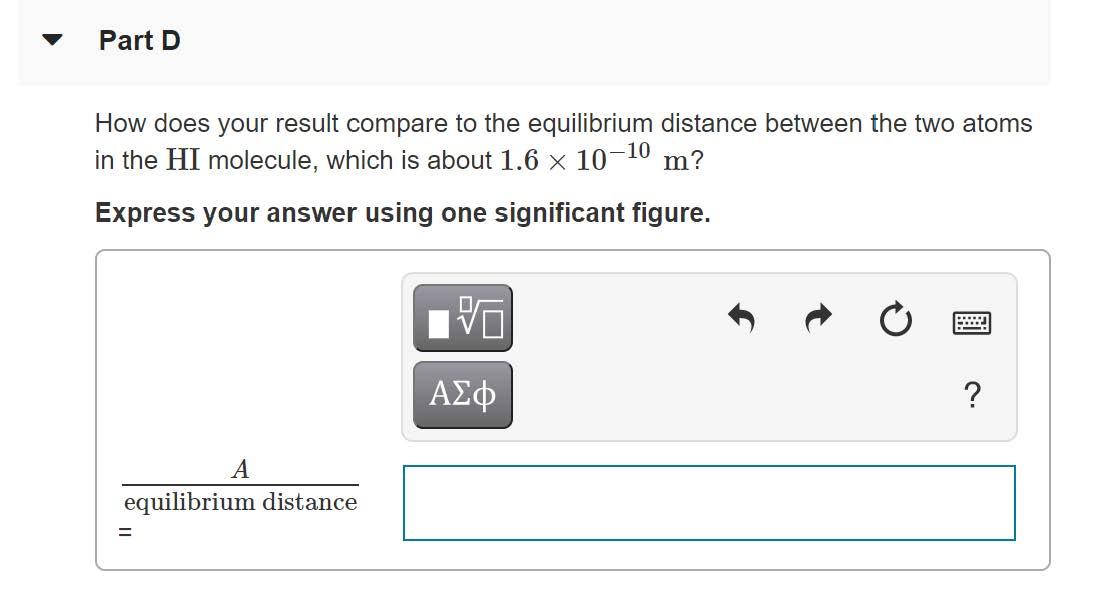
Quantum mechanics is used to describe the vibrational motion of molecules, but analysis using classical physics gives some useful insight. In a classical model the vibrational motion can be treated as SHM of the atoms connected by a spring. The two atoms in a diatomic molecule vibrate about their center of mass, but in the molecule HI, where one atom is much more massive than the other, we can treat the hydrogen atom as oscillating in SHM while the iodine atom remains at rest. Part A A classical estimate of the vibrational frequency is f = 7.0 x 1013 Hz. The mass of a hydrogen atom differs little from the mass of a proton. If the HI molecule is modeled as two atoms connected by a spring, what is the force constant of the spring? Express your answer to two significant figures and include the appropriate units. HA k = Value Units Part B The vibrational energy of the molecule is measured to be about 5 x 10-20 J. In the classical model, what is the maximum speed of the H atom during its SHM? Express your answer to one significant figure and include the appropriate units. HẢ ? Value Units V = Part C What is the amplitude of the vibrational motion? Express your answer to one significant figure and include the appropriate units. HA ? A = Value Units %3D Part D How does your result compare to the equilibrium distance between the two atoms in the HI molecule, which is about 1.6 x 10-10 m? Express your answer using one significant figure. ? A equilibrium distance Quantum mechanics is used to describe the vibrational motion of molecules, but analysis using classical physics gives some useful insight. In a classical model the vibrational motion can be treated as SHM of the atoms connected by a spring. The two atoms in a diatomic molecule vibrate about their center of mass, but in the molecule HI, where one atom is much more massive than the other, we can treat the hydrogen atom as oscillating in SHM while the iodine atom remains at rest. Part A A classical estimate of the vibrational frequency is f = 7.0 x 1013 Hz. The mass of a hydrogen atom differs little from the mass of a proton. If the HI molecule is modeled as two atoms connected by a spring, what is the force constant of the spring? Express your answer to two significant figures and include the appropriate units. HA k = Value Units Part B The vibrational energy of the molecule is measured to be about 5 x 10-20 J. In the classical model, what is the maximum speed of the H atom during its SHM? Express your answer to one significant figure and include the appropriate units. HẢ ? Value Units V = Part C What is the amplitude of the vibrational motion? Express your answer to one significant figure and include the appropriate units. HA ? A = Value Units %3D Part D How does your result compare to the equilibrium distance between the two atoms in the HI molecule, which is about 1.6 x 10-10 m? Express your answer using one significant figure. ? A equilibrium distance
Expert Answer:

Related Book For 

Numerical Methods for Engineers
ISBN: 9780071244299
5th Edition
Authors: Steven C. Chapra, Raymond P. Canale
Posted Date:
Students also viewed these physics questions
-
What manufacturing cost term is used to describe the cost of materials that are an integral part of the manufactured end product?
-
The sales life cycle is used to describe the phases a product goes through from introduction to withdrawal from the market. The four phases are: (a) introduction, (b) growth, (c) maturity, and (d)...
-
What are five descriptive statistics used to describe the basic properties of variables?
-
The compound bow in the previous problem requires 152 J of work to draw the string back to x = 0.540 m, at which point the archer need only exert 250 N of force to hold the string in place. (a)...
-
What are the main criticisms of RBC theory and how do its supporters defend it?
-
Presented below is information related to Lor Co. 1. On April 5, purchased merchandise from Garcia Company for $19,000, terms 2/10, net/30, FOB shipping point. 2. On April 6, paid freight costs of...
-
Leicht Transfer & Storage provides warehousing services and often purchases pallets from Pallet Central. The companies followed a standard practice for documenting these transactions in which Pallet...
-
What company policies or procedures would you recommend to prevent each of the following activities? a. A clerk at the Paul Yelverton Company faxes a fictitious sales invoice to a company that...
-
When President Obama was President he had discussed raising income taxes for individuals earning over $250,000 in income. Explain how these higher income taxes will affect the aggregate demand curve....
-
Joy Wu, a PA, is planning her first audit of a closely held small business. In prior years, Wu compiled the financial statements of the company. She also helped to set up its accounting system and...
-
During the month of February 2021, the following transactions occurred: Feb. 1 Collected P118,000 from customers on account 2 Paid P68,000 of accounts due less discounts of 2% 4 Purchased...
-
Larsen & Toubro Ltd (www.larsentoubro.com) is an Indian multinational conglomerate listed on the Bombay Stock Exchange. In the financial year 2011, the companys net income was 4,456 crore, EPS was...
-
The rates of returns data from year 2000 to 2009 in Table 4.15 is listed below: At the = 0.05, can we conclude that the two stocks and the general market generate about the same average rate of...
-
A chicken farm came up with 4 different ways of mixing chicken feeds. The feeds were tested on 20 chickens. The results, given in terms of the chickens weight, are presented in the accompanying...
-
For these problems, use any web service that provides financial information. Good examples are Yahoo! Finance, Hemscott, Reuters and FT.com. You can also go to the companys website and download...
-
The performances of 250 salespeople in a company are summarized in the following table. Derive the expected frequencies, assuming that the data are from a normal distribution. Do the data collected...
-
Give a tight bound of the nearest runtime complexity class for each of the following code fragments in Big-Oh notation, in terms of the variable N. In other words, write the code's growth rate as N...
-
Write a paper detailing a geographic information system (GIS) of your own design that would utilize data in an original manner.
-
Given the equations 2x1 6x2 x3 = 38 3x1 x2 + 7x3 = 34 8x1 + x2 2x3 = 20 (a) Solve by Gauss elimination with partial pivoting. Show all steps of the computation. (b) Substitute your results...
-
Use the shooting method to solve d 2 T / dx 2 1x l0 -7 ( T + 273) 4 + 4(l50 T ) = 0 Obtain a solution for boundary conditions: T (0) = 200 and T (0.5)= 100.
-
Use Romberg integration to evaluate To an accuracy of s = 0.5%. Your results should be presented in the form ofFigure. sinx dx 1+x2
-
Suppose you are the financial advisor to a firm that is in good financial health. What suggestions would you make to the firms management about obtaining borrowed funds if both financially healthy...
-
Use a core principle from Chapter 1 to explain why, everything else being equal, a software company might find it more expensive to issue debt than a furniture store?
-
Financial intermediaries specialize in reducing costs by: a. Pooling the resources of small savers and lending them to large borrowers. b. Providing safekeeping, accounting services, and access to...

Study smarter with the SolutionInn App


