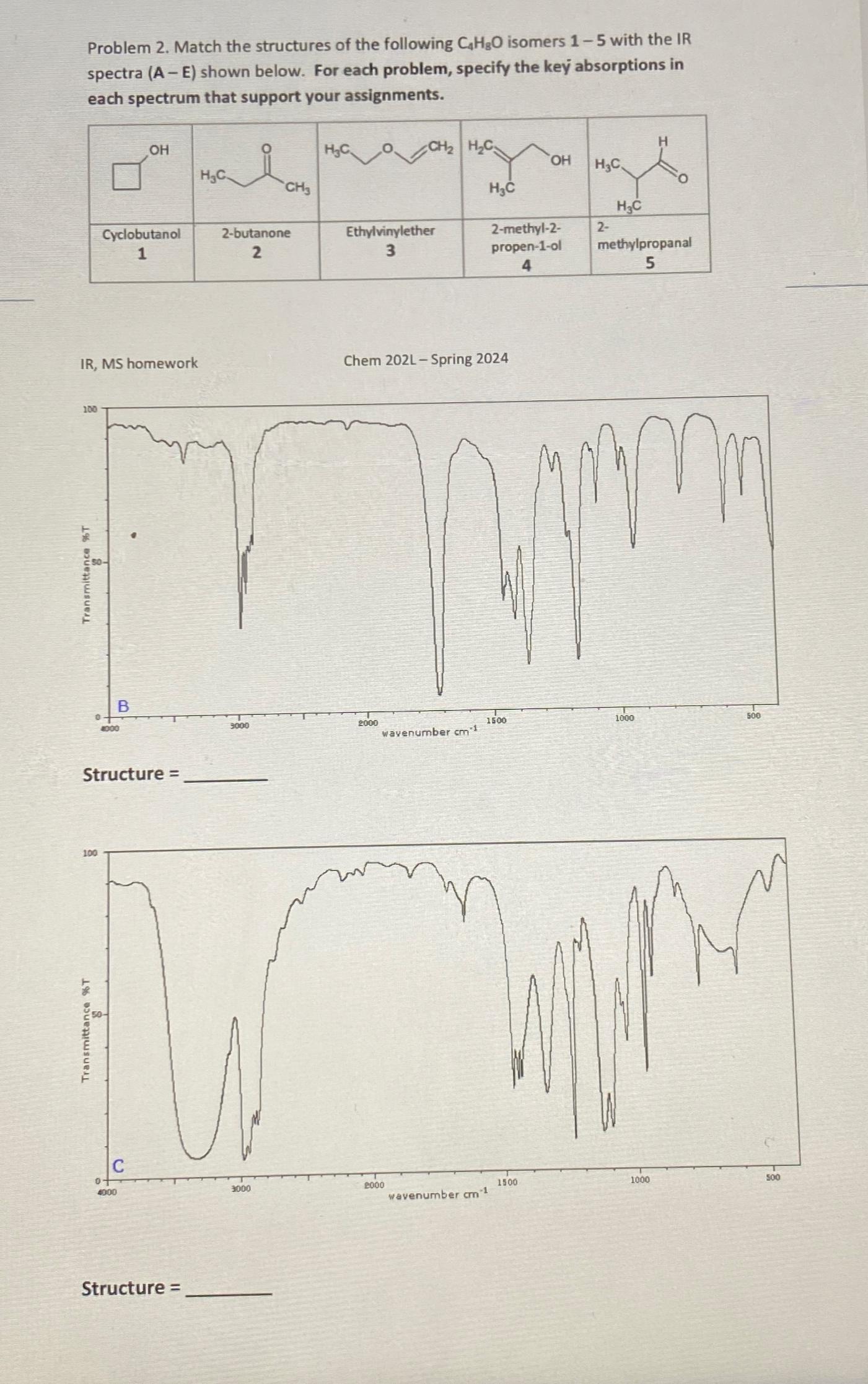
Problem 2. Match the structures of the following CHO isomers 1-5 with the IR spectra (A-E)...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Problem 2. Match the structures of the following CHO isomers 1-5 with the IR spectra (A-E) shown below. For each problem, specify the key absorptions in each spectrum that support your assignments. 100 IR, MS homework Transmittance %T 0 100 Transmittance %T Cyclobutanol 1 4000 Structure = 0 OH B 4000 Structure = HC i 2-butanone 2 9000 CH3 3000 HC Ethylvinylether 3 CH HC 2000 Chem 202L - Spring 2024 wavenumber cm 1 2000 HC wavenumber cm-1 2-methyl-2- propen-1-ol 4 1500 OH 1500 HC 2- HC methylpropanal 5 mm 1000 1000 500 500 Problem 2. Match the structures of the following CHO isomers 1-5 with the IR spectra (A-E) shown below. For each problem, specify the key absorptions in each spectrum that support your assignments. 100 IR, MS homework Transmittance %T 0 100 Transmittance %T Cyclobutanol 1 4000 Structure = 0 OH B 4000 Structure = HC i 2-butanone 2 9000 CH3 3000 HC Ethylvinylether 3 CH HC 2000 Chem 202L - Spring 2024 wavenumber cm 1 2000 HC wavenumber cm-1 2-methyl-2- propen-1-ol 4 1500 OH 1500 HC 2- HC methylpropanal 5 mm 1000 1000 500 500
Expert Answer:
Answer rating: 100% (QA)
What you have presented is an infrared IR spectroscopy matching problem You need to match the given IR spectra AE to the correct structures of the C4H... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
The following spectra for A and B correspond to two structural isomers. The NMR singlet at (1.16 in spectrum A disappears when the sample is shaken with D2O.The singlet at (0.6 ppm in the spectrum of...
-
The magnet has mass 3 . 8 5 kg and the force pulling it to the right is 1 3 6 . 8 N . When the magnet hits the floor, it continues being pulled to the right by the same magnetic force as before. The...
-
Assume these simple exchange rate relationships to answer the questions below: Forward rate equals the expected spot rate. The interest differential between two countries must be equal to the...
-
Complete each statement. We find the quotient of two monomials using the _____ rule for _____ .
-
George Oppenheimer, an agent for Wellington Farms of Massachusetts, Inc., had contacted Mark Kiriakou from the Capital Area Food Bank regarding an order for frozen turkey meat. In an exchange of...
-
Presented below is the trial balance of Scott Butler Corporation at December 31, 2014. Instructions Prepare a balance sheet at December 31, 2014, for Scott Butler Corporation. (Ignore incometaxes.)...
-
ESG CSR Study caseSunset Manufacturing Inc. manufactures Umbrellas in the Western Canadian market, and is comparing the budget for the current year with the actual results. The market has been good...
-
Financial statement data of ABC Fencing, Inc. include the following items: Cash .................... $ 21,000 Short-Term Investments .......... 25,000 Accounts Receivable, Net .......... 102,000...
-
Q2 Draw the S.F. and B.M. diagrams for the beam loaded as shown in Fig below, and determine (a) (b) The position and magnitude of the maximum B.M., and The position of point of contra flexure, if any...
-
The following is the residual momentum observer derivation. Robot dynamics is given as follows Define momentum: p = M(q)q M + Cq + g = t+r Compute p using t, Text, g, C, and q
-
Give the exact parent peak mass of the compound C6H6C16. Assume that ONLY the most common isotope is present. Give the mass to 4 decimal places. The masses (in Daltons) of selected isotopes and their...
-
Briefly describe what rethinking the employee experience of remote workers is, along with how and why it will have the most significant impact on the workplace. Then, explain how it could be applied...
-
An ideal monatomic gas expands isothermally from 0.550 m to 1.25 m at a constant temperature of 760 K. If the initial pressure is 1.11 x 105 Pa find the following. (a) the work done on the gas -54400...
-
1. Use a stack to determine if the expression's parentheses are balanced. develop a stack trace like in our lecture (and page 360) that shows each step using a series of boxes....
-
Assume you have the following jobs to execute with one processor: Note: 1: highest; 5: lowest Process Arival CPU Burst Priority Time time 35 4 B 25 15 15 30 15 E 40 15 1 Using a preemptive priority...
-
Which one of the following anhydrous chloride is not obtained on direct heating of its hydrated chloride? (A) BaCl2 (B) CaClz (C) MgCl2 (D) SrCl2
-
Predict the sites on each of the following compounds where the reaction can occur: a. CH3CH==CHOCH3 + H+ b. Cl
-
What carbonyl compounds are required to prepare a compound with molecular formula C10H10Owhose spectrum is shown? (ppm)
-
List the following compounds in order of decreasing Keq for hydrate formation: C CH3 C CH3 O,N
-
Go to the Web site for the Software Engineering Institute of Carnegie Mellon University at https://resources.sei.cmu.edu/asset_files/SpecialReport/1994_003_001_16265.pdf and access the software...
-
The majority of the project budget is expended upon: a. Project plan development. b. Project plan execution. c. Project termination. d. Project communication.
-
Which of the following is the most critical component of the triple constraint? a. Time, then cost, then quality. b. Quality, then budget, then time. c. Scope. d. They are all of equal importance...

Study smarter with the SolutionInn App


