Q : Calculate the standard cell potentials of galvanic cells in which the following reactions take place:
Fantastic news! We've Found the answer you've been seeking!
Question:
Q :
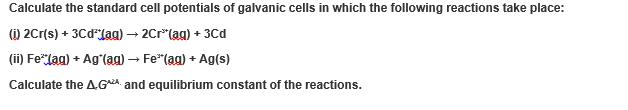
Transcribed Image Text:
Calculate the standard cell potentials of galvanic cells in which the following reactions take place: (i) 2Cr(s) + 3CdJag) → 2Cr(ag) + 3Cd (ii) Fe* (ag) + Ag (ag) → Fe*(ag) + Ag(s) Calculate the AGA and equilibrium constant of the reactions. Calculate the standard cell potentials of galvanic cells in which the following reactions take place: (i) 2Cr(s) + 3CdJag) → 2Cr(ag) + 3Cd (ii) Fe* (ag) + Ag (ag) → Fe*(ag) + Ag(s) Calculate the AGA and equilibrium constant of the reactions.
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
P Ltd has two divisions, Q and R, that operate as profit centres. Division Q has recently been set up to provide a component (Comp1) which division R uses to produce its product (ProdX). Prior to...
-
1. The following relations describe demand and supply. Q = 700 - 100P (1) Q = -100 + 100P (2) Where P is price in dollar and Q is quantity in unit. a. Which equation (1) or (2), is demand equation...
-
Let Q be a set of n points in the plane. We say that point (x, y) dominates point (x?, y?) if x ? x? and y ? y?. A point in Q that is dominated by no other points in Q is said to be maximal. That Q...
-
An undamped mass spring system is released from rest from an initial displacement of x = 0.24 m and starts to oscillate. You see that the mass reaches its largest positive displacement for the first...
-
Compute the impact on the money multiplier of an increase in the currency-to-deposit ratio from 10 percent to 15 percent when the reserve requirement is 10 percent of deposits, and banks desired...
-
Suppose Alexandra Pater, the manager of the Food and Lodging Division at Winter Wonderland Resort, has hired you as a consultant to help her examine her division's performance under several different...
-
Aaron Reed, a photographer, was in a dispute with Ezelle Investment Properties, Inc., over Ezelle allegedly using one of Reeds photographs without permission. Reed sent Ezelle a cease-and-desist...
-
Converting from Modified Accrual to Accrual Accounting. The Village of Rodale keeps its governmental find accounting records on a modified accrual basis. At the end of the fiscal year, the village...
-
Objectives: Your company is currently planning to automate many different system administration tasks. Your manager has asked you to research a system administration task and implement it using...
-
Each salesperson at Rembrandt Auto-Mart is assigned an ID number that consists of five characters. The first three characters are numbers. The fourth character is a letter: either the letter N if the...
-
Show how to find the transfer function by using manual methods and reading it graphical. An estimate is sufficient. Step Response 0.6 0.5 0.4 0.3 0.2 0.1 3 4 7 9. 10 Time (seconds) 8. 2. Amplitude
-
Suppose your model predicts that overweight children have an 80% higher risk of suffering from diabetes in their adult life. If data show that overweight children do not suffer from diabetes as...
-
Suppose that in a given economy all goods and services produced are sold in perfectly competitive markets. Would you represent this economy using the classical or Keynesian approach? Explain why.
-
Suppose Japan has a GDP of $5 trillion, and that its national savings rate is 25%. Assuming Japan is an open economy, a) calculate Japans investment if net exports are 1% of GDP. b) calculate Japans...
-
Social media firms strategy has three pillars: (1) keeping user addicted to the content, (2) collecting data about user behavior, (3) selling advertisements targeting specific users. Do you really...
-
Comment on the effect of a decrease in autonomous investment on wealth when the economy can be considered a a) closed economy. b) small open economy.
-
What is the expected return of the three-stock portfolio described below? Common Stock Alko Inc. Belmont Co. Cardo Inc. Market Value 25.000 100,000 75,000 Expected Return 38% 10% 16%
-
At the beginning of the year, Lam Ltd. had total assets of $800,000 and total liabilities of $500,000. Use this information to answer each of the following independent questions. (a) If Lam's total...
-
(a) Define the terms limiting reactant and excess reactant. (b) Why are the amounts of products formed in a reaction determined only by the amount of the limiting reactant? (c) Why should you base...
-
The iodine bromide molecule, IBr, is an inter-halogen compound. Assume that the molecular orbitals of IBr are analogous to the homo nuclear diatomic molecule F2. (a) Which valence atomic orbitals of...
-
The figure shows the three lowest regions of Earth's atmosphere. (a) Name each and indicate the approximate elevations at which the boundaries occur. (b) In which region is ozone a pollutant? In...
-
Use the Internet to access Home Depot's most recent annual report. a. What evidence did you find that Home Depot continues to rely on present worth analysis in its corporate decision making? b. Does...
-
Use an Internet search engine to identify three companies or agencies that use present worth analysis to make business decisions as evidenced in their annual reports.
-
Use the Internet to access ConocoPhillips's most recent annual report. a. What evidence do you find that ConocoPhillips continues to rely on present worth analysis in its corporate decision making?...

Study smarter with the SolutionInn App



