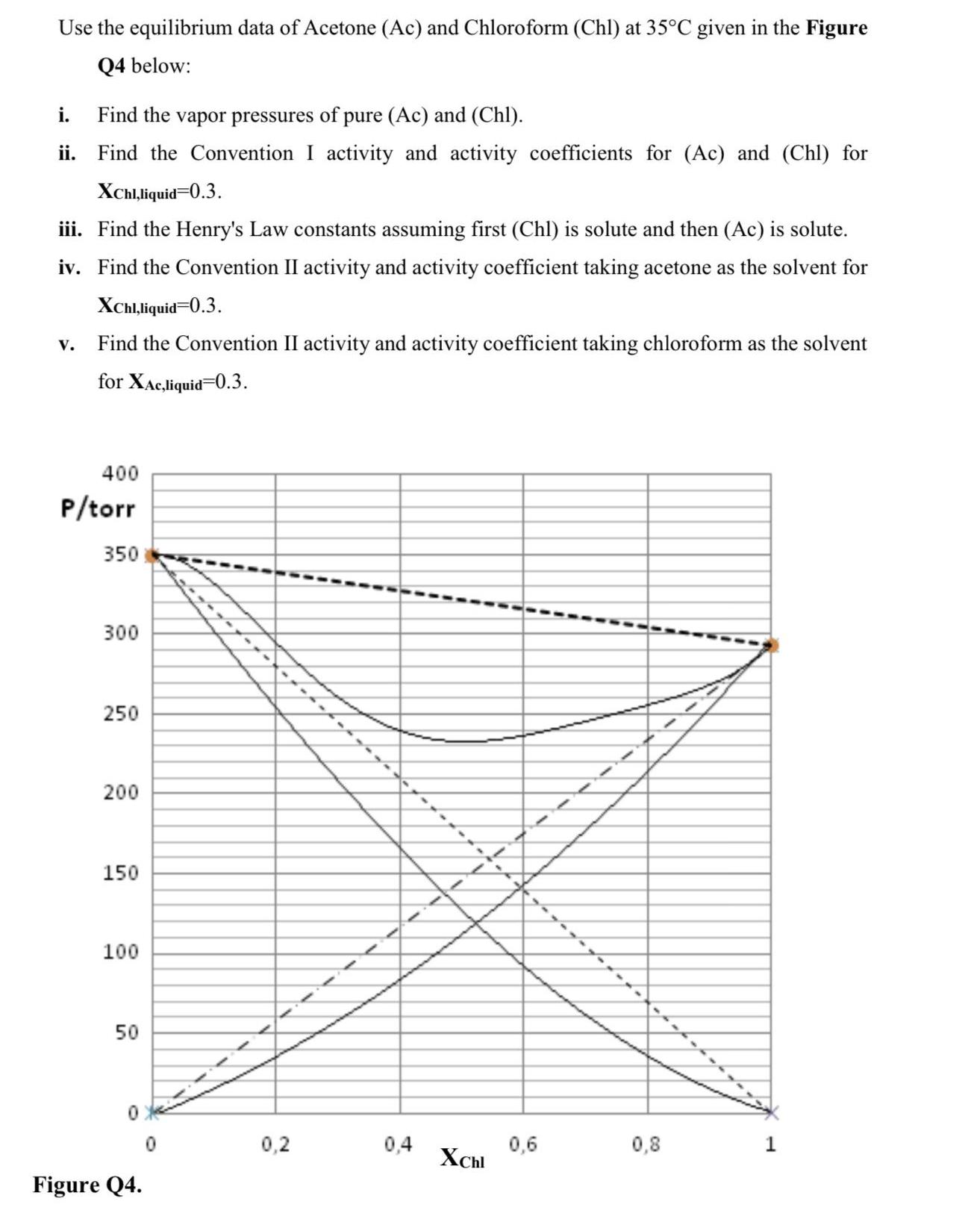
Use the equilibrium data of Acetone (Ac) and Chloroform (Chl) at 35C given in the Figure...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Use the equilibrium data of Acetone (Ac) and Chloroform (Chl) at 35°C given in the Figure Q4 below: i. Find the vapor pressures of pure (Ac) and (Chl). ii. Find the Convention I activity and activity coefficients for (Ac) and (Chl) for XChl,liquid=0.3. iii. Find the Henry's Law constants assuming first (Chl) is solute and then (Ac) is solute. iv. Find the Convention II activity and activity coefficient taking acetone as the solvent for XChl,liquid=0.3. V. Find the Convention II activity and activity coefficient taking chloroform as the solvent for XAc,liquid=0.3. 400 P/torr 350 300 250 200 150 100 50 Figure Q4. 0,2 0,4 XChl 0,6 0,8 Use the equilibrium data of Acetone (Ac) and Chloroform (Chl) at 35°C given in the Figure Q4 below: i. Find the vapor pressures of pure (Ac) and (Chl). ii. Find the Convention I activity and activity coefficients for (Ac) and (Chl) for XChl,liquid=0.3. iii. Find the Henry's Law constants assuming first (Chl) is solute and then (Ac) is solute. iv. Find the Convention II activity and activity coefficient taking acetone as the solvent for XChl,liquid=0.3. V. Find the Convention II activity and activity coefficient taking chloroform as the solvent for XAc,liquid=0.3. 400 P/torr 350 300 250 200 150 100 50 Figure Q4. 0,2 0,4 XChl 0,6 0,8
Expert Answer:
Answer rating: 100% (QA)
To find the various properties and coefficients as requested we can use the given equilibrium data for Acetone Ac and Chloroform Chl at 35C It appears ... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these chemical engineering questions
-
Use the graph input tool to help you answer the following questions. You will not be graded on any changes you make to this graph. Note: Once you enter a value in a white field, the graph and any...
-
The revenue R (in millions of dollars) for a beverage company is related to its advertising expense by the function R = 1/100,000 (-x3 + 600x2), 0 ¤ x ¤ 400 Where x is the amount spent...
-
The "Optimal Deadbeat" Problem: The World Bank is considering a stream of loans to the Puglian government to help it develop its nationalized oil fields and refineries. This is the only set of loans...
-
The director of cost management for Odessa Company uses a statistical control chart to help management determine when to investigate variances. The critical value is 1 standard deviation. The company...
-
Consider a finite-duration sequence x[n] of length p such that x[n] = 0 for n < 0 and n P. We want to compute samples of the Fourier transform at the N equally spaced frequencies k = 2k/N, k = 0,...
-
What is the direction of the magnetic field lines inside the bar magnet of Figure 27.13? Figure 27.13 Magnetic field line pattern surrounding a bar magnet. N S
-
Refer to the data for Midway Cycles in E5-19A. Requirements 1. Compute the cost of goods sold under FIFO. 2. Compute the cost of goods sold under LIFO. 3. Which method results in the higher cost of...
-
The following is the unadjusted trial balance for Rainbow Lodge Ltd. at its year end, May 31, 2015. The company adjusts its accounts monthly. Additional information: 1. An annual insurance policy was...
-
Some families have been impacted by the pandemic to draw members back more close to home whilst other families seem more than ever to want their offspring to build careers abroad. Can yu please...
-
Amy Lloyd is interested in leasing a new Honda and has contacted three automobile dealers for pricing information. Each dealer offered Amy a closed-end 36-month lease with no down payment due at the...
-
Changing a learning culture or an learned work culture can be very difficult. In the past 5 years we a needed to have a culture shift away from the arrest model and to a program...
-
1) The tensile test has been applied to a 6150 Steel specimen. The dimensions and properties are given below: Specimen diameter: Peak load: Extension at peak load: Strain at peak load: Load at onset...
-
Assume a price decrease from currently 5.00 to 3.00 for your product. Calculate the price elasticity of demand. Is the demand elastic or inelastic?
-
What strategies do operating systems use for resource allocation to prevent resource starvation, and how do they mitigate the risks of resource hoarding by individual processes ?
-
1. Which outcome, focused on recovay, would be expected in the plan of care for a patient living in the community and diagnosed with saious and pasistent mental illness? Within 3 months, the patient...
-
How would you explain the formula for computing individual taxable income. Why do you think this formula is so much more complicated than the formula for computing corporate taxable income? Explain...
-
Solve this problem in C++. Imagine you have the Queue class from your CSE225 lecture. Create a queue (any size) in the main function. Enqueue a few items (user input or random or hard coded). Then...
-
The Place-Plus real estate development firm in Problem 24 is dissatisfied with the economists estimate of the probabilities of future interest rate movement, so it is considering having a financial...
-
A frustrated professor once claimed that if all the reports she had graded in her career were stacked on top of one another, they would reach from the Earth to the moon. Assume that an average report...
-
In terms of Le Chateliers Principle, explain why steam reforming is done at low pressures while methanol synthesis is performed at moderate to high pressures. Explain why reforming is done at high...
-
The Cookenwythe Gas Company pumps propane gas to the nearby Noxious Chemicals, Inc., polypropylene production plant. The gas is metered at the noxious plant at 400m 3 /h at 4.7atm gauge and 30C. The...
-
What is the effect of pressure on equilibrium conversion of a gas-phase chemical reaction?
-
The diathermal wall (a) Is incapable of exchanging heat with the surroundings (b) Permits the full flow of heat from the system to the surroundings and vice versa (c) Both (a) and (b) (d) None of...
-
The total energy of a system comprises (a) Kinetic energy, potential energy and vibrational energy (b) Kinetic energy, potential energy and rotational energy (c) Kinetic energy, potential energy and...

Study smarter with the SolutionInn App


