Question 1 1 pts How many iatoms are in 45 moles of L O GAS atoms...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
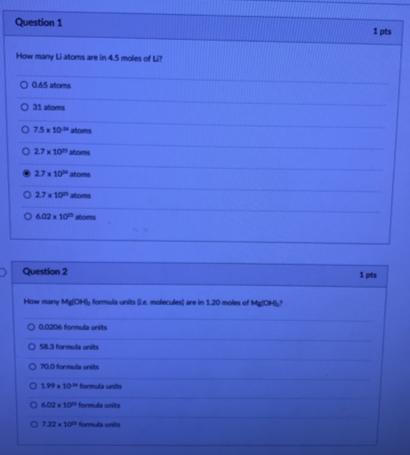
Question 1 1 pts How many iatoms are in 45 moles of L O GAS atoms O 31 atoms O 75x 10atoms O 27x 10atoms 27 10 atome O 27 10atome O 402x 10 oms Question 2 1 pts How many MOH, launtsa mdee rein1.20 mol of MON 6 aa0e oma urts O S enits O 00 ds ns 0 19 10 Question 1 1 pts How many iatoms are in 45 moles of L O GAS atoms O 31 atoms O 75x 10atoms O 27x 10atoms 27 10 atome O 27 10atome O 402x 10 oms Question 2 1 pts How many MOH, launtsa mdee rein1.20 mol of MON 6 aa0e oma urts O S enits O 00 ds ns 0 19 10
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
How many atoms are present in 4.55 mol of Fe?
-
How many atoms are present in 0.0665 mol of K?
-
How many moles are present in 334 L of H2 at STP?
-
Which of the following statements about Visa credit card sales is incorrect? (a) The credit card issuer makes the credit investigation of the customer. (b) The retailer is not involved in the...
-
Calculate Altmans Z score for Home Depot in fiscal year 2011 (as of January 29, 2012) and then compare it with Moodys rating chart. Is this company an IG or non-IG? All numbers are inmillions....
-
Make a flow chart algorithm using start/end = circle, process/task = rectangle, diamond= decision, arrow = flow of control, input/ output = parallelogram, to make a pb/j sandwhich.
-
Consider the regression model in Problem 3.43. The value of the adjusted $R^{2}$ is Data From Problem 3.43 You have fit a linear regression model with three predictors to a sample of 50 observations....
-
Customers checking out at Food Tiger arrive in a single-line queue served by two cashiers at a rate of eight per hour according to a Poisson distribution. Each cashier processes customers at a rate...
-
The y-intercept of the graph of the exponential function f(x) = 7.5(0.98)* is. Answer:
-
1. What are the advantages and disadvantages of the in-home method of selling Project Home products? 2. What other channels of distribution might Project Home use? 3. What do you think about the name...
-
Benefits Your company provides traditional benefits program. The specifics are as follows: a. Health insurance: the organization contributes $300 per employee per month, which covers the cost for the...
-
What is the value of $250 at 6% interest rate in one year?
-
Description Why might an organization's legal rules sometimes be insufficient to fulfilling one's ethical responsibilities? Can you think of cases in which a businessperson had done something legally...
-
Shopping Mall contracts with Gardener to do landscaping work for $30,000. The next lowest bid for the work had been $45,000. Mall pays $15,000 of the total price up front. After doing a substantial...
-
Carol, a resident of Denver, Colorado, is a homemaker who has a private collection of old China. She sees a plate advertised on eBay by a Canadian seller, Antiques-R-Us, which is located in Toronto,...
-
Teacher owns a school that offers tutoring in writing, and he contracts with Printer for 1,000 brochures with a headline that states, "Writing for Success!" The brochures are to be delivered in...
-
An anaerobic digester treating brewery wastewater produces 17,500 m of dry biogas measured at STP conditions per day. Dry here means moisture-free biogas. The methane (CH4) content of the dry biogas...
-
A report from the college dean indicates that for the previous semester, the grade distribution for the Department of Psychology included 135 As, 158 Bs, 140 Cs, 94 Ds, and 53 Fs. Determine what kind...
-
In which of the following are the species written in decreasing order by size of radius? (a) Be, Mg, Ba, (b) N3-, O2-, F-, (c) Tl3+, Tl2+, Tl+.
-
Barium metal crystallizes in a body-centered cubic lattice (the Ba atoms are at the lattice points only). The unit cell edge length is 502 pm, and the density of the metal is 3.50 g/cm3. Using this...
-
Progesterone is a hormone responsible for female sex characteristics. In the usual shorthand structure, each point where lines meet represent a C atom, and most H atoms are not shown. Draw the...
-
Who was Phar-Mors flamboyant Chief Executive Officer?
-
Which of the following generally is not considered something of value? 1. Cash, money or checks 2. Airline miles or hotel credits associated with frequent activity (e.g., frequent flier miles) 3. An...
-
Which of the following is not one of the five major categories of fraudulent disbursements? 1. Payroll schemes 2. Expense reimbursement schemes 3. Shell company schemes 4. Billing schemes

Study smarter with the SolutionInn App



