Question 1: Phase Diagrams (10 points) The Cu-Zn (brass) phase diagram is given below. Use it...
Fantastic news! We've Found the answer you've been seeking!
Question:
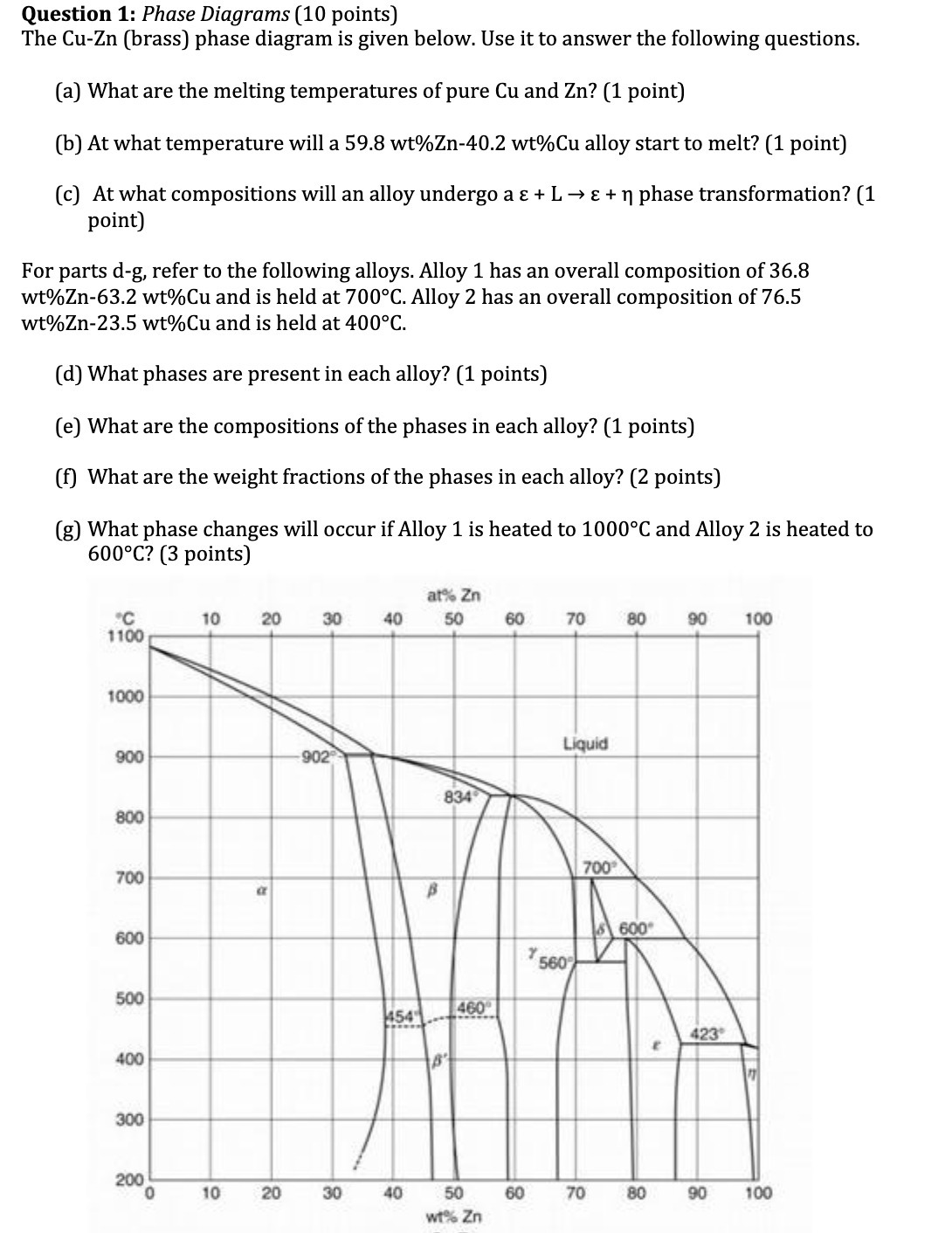
Transcribed Image Text:
Question 1: Phase Diagrams (10 points) The Cu-Zn (brass) phase diagram is given below. Use it to answer the following questions. (a) What are the melting temperatures of pure Cu and Zn? (1 point) (b) At what temperature will a 59.8 wt% Zn-40.2 wt%Cu alloy start to melt? (1 point) (c) At what compositions will an alloy undergo a + L + phase transformation? (1 point) For parts d-g, refer to the following alloys. Alloy 1 has an overall composition of 36.8 wt%Zn-63.2 wt%Cu and is held at 700C. Alloy 2 has an overall composition of 76.5 wt%Zn-23.5 wt%Cu and is held at 400C. (d) What phases are present in each alloy? (1 points) (e) What are the compositions of the phases in each alloy? (1 points) (f) What are the weight fractions of the phases in each alloy? (2 points) (g) What phase changes will occur if Alloy 1 is heated to 1000C and Alloy 2 is heated to 600C? (3 points) C 1100 1000 900 800 700 600 500 400 300 200 0 10 10 20 a 20 30 -902 30 40 454 40 at% Zn 50 B 834 B 460 50 wt% Zn 60 60 Y 70 Liquid 560 700 70 80 600 80 E 90 100 423 90 100 Question 1: Phase Diagrams (10 points) The Cu-Zn (brass) phase diagram is given below. Use it to answer the following questions. (a) What are the melting temperatures of pure Cu and Zn? (1 point) (b) At what temperature will a 59.8 wt% Zn-40.2 wt%Cu alloy start to melt? (1 point) (c) At what compositions will an alloy undergo a + L + phase transformation? (1 point) For parts d-g, refer to the following alloys. Alloy 1 has an overall composition of 36.8 wt%Zn-63.2 wt%Cu and is held at 700C. Alloy 2 has an overall composition of 76.5 wt%Zn-23.5 wt%Cu and is held at 400C. (d) What phases are present in each alloy? (1 points) (e) What are the compositions of the phases in each alloy? (1 points) (f) What are the weight fractions of the phases in each alloy? (2 points) (g) What phase changes will occur if Alloy 1 is heated to 1000C and Alloy 2 is heated to 600C? (3 points) C 1100 1000 900 800 700 600 500 400 300 200 0 10 10 20 a 20 30 -902 30 40 454 40 at% Zn 50 B 834 B 460 50 wt% Zn 60 60 Y 70 Liquid 560 700 70 80 600 80 E 90 100 423 90 100
Expert Answer:

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these mechanical engineering questions
-
"internet radios" for streaming audio, and personal video recorders and players. Describe design and evaluation processes that could be used by a start-up company to improve the usability of such...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Suppose we have an automatic module on the module path named lizard-^-cricket-^-1.0.0-SNAPSHOT.jar and no Automatic-Module-Name specified. What module name should named modules use to reference it?...
-
Yello Bus Lines uses the units-of-activity method in depreciating its buses. One bus was purchased on January 1, 2019, at a cost of $148,000. Over its 4-year useful life, the bus is expected to be...
-
In Problems 1 to 3. determine whether each matrix A is diagonalizable. If it is, determine a matrix P that diagonalizes it and compute P-1 AP. You can obtain P-1 AP directly from careful construction...
-
Identify the four components of an ecosystem. After you do this try to visualize the interactions of these four components of an ecosystem as illustrated by Figure 2. 2 in the textbook. Does this...
-
1. Irad Liu, of Commerce, Texas, is in the 25 percent marginal tax bracket and is considering the tax consequences of investing $2000 at the end of each year for 30 years in a tax-sheltered...
-
Find the two unknown sides in this triangle. Keep 4 significant digits. x+3.10m -240m-x 5.21m
-
Wild (2016) states that Predictable variations in demand may cause an operation some problems, but at least if demand variation is known, the operation can plan for it. Unpredictable variation in...
-
23. Using Kirchoff's loop and junction rules we obtain these equations for the currents in 21 +21-5=0 different parts of the circuit. Find I and 1. 4-41-0
-
A series RLC combination that has L = 2.0 H, C =2.0 F, and R = 20.092 is driven by a generator that has a maximum emf of 100.0V and a frequency which can be varied. Find: (a) the resonate frequency...
-
3. Determine whether the following graph is bipartite. If it is, give the bipartition sets. If not, explain why not (you need to explain your answer to get full marks!) [5 points]
-
Given the two time series P = [2, 1, 4, 10] and Q = [2, 5, 2, 7]. Fill the distance matrix M from the bottom left corner according to the formula: M(i, j)= |x, yj|+ min(M(i-1, j-1), M(i, j-1), M(i -...
-
2. Consider the following procedure, which takes a list L, ts length n < and a positive integer l as parameters function MYSTERY(L, n, l) for i from 1 to n - l + 1 do for j from i+ 1 to n-l+ 1 do...
-
We drop a needle of length I on a ruled paper with lines separated by a distance d. The location of drop and the angle are independent and uniformly random. (a) What is the probability of the needle...
-
Proposals have been made to ?sail? spacecraft to the outer solar system using the pressure of sunlight, or even to propel interstellar spacecraft with high-powered, Earth-based lasers. Sailing...
-
Detonation of nitroglycerin proceeds as follows: (a) If a sample containing 2.00 mL of nitroglycerin (density 1.592 g/mL is detonated, how many total moles of gas are produced? (b) If each mole of...
-
The solubility of Cr(NO3)3 ( 9 H2O in water is 208 g per 100 g of water at 15oC. A solution of Cr(NO3)3 ( 9 H2O in water at is formed by dissolving 324 g in 100 g water. When this solution is slowly...
-
(a) What is the mass percentage of iodine (I2) in a solution containing 0.035 mol I2 in 125 g of CCl4? (b) Seawater contains per 0.0079 g Sr2+ kilogram of water. What is the concentration of Sr2+...
-
Find the Laplace transform of the function \[y(t)=\alpha \sin ^{2} t+\beta \cos ^{2} t .\]
-
Consider the dynamic system, a mass, spring and damper structure, discussed in Example 2.1 and shown in Figure 2.9. Find the Transfer Function model of the dynamic system, where \(f(t)\) is the input...
-
Consider a system with the following State-Variable Matrix model where \(x_{1}(t)\) and \(x_{2}(t)\) are the state variables, the \(u(t)\) is the input and \(y(t)\) is the output. Find the Transfer...

Study smarter with the SolutionInn App


