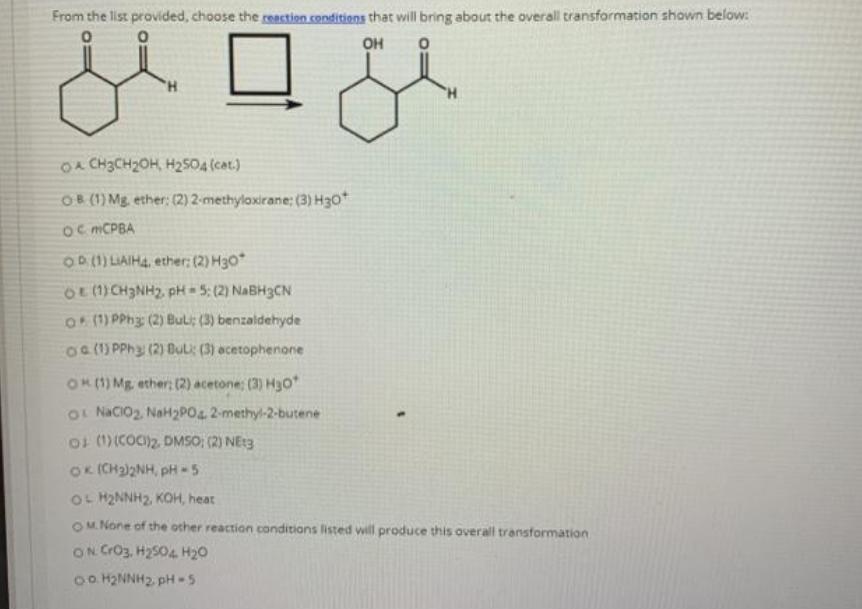
From the list provided, choose the reastion conditions that will bring about the overall transformation shown...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
From the list provided, choose the reastion conditions that will bring about the overall transformation shown below: он OA CH3CH2OH, H2504 (cat.) OB (1) Mg. ether: (2) 2-methyloxirane; (3) H30 Oc MCPBA OD (1) LIAIH4, ether: (2) H30* OE (1) CH3NH2, pH = 5: (2) NABH3CN O ) PPh3 (2) Buli: (3) benzaldehyde Oa(1) PPH3 (2) ĐuL: (3) acetophenone OK() Mg, ether; (2) acetone; (3) H30 OL NaCIO2, NaH2PO4 2-methyl-2-butene ot (1)(COC)2. DMSO; (2) NEt3 OK (CH2)2NH, pH - 5 OL H2NNH2, KOH, heat OM None of the other reaction conditions listed will produce this overall transformation ON CrO3, H2504 H20 o. H2NNH2, pH -5 From the list provided, choose the reastion conditions that will bring about the overall transformation shown below: он OA CH3CH2OH, H2504 (cat.) OB (1) Mg. ether: (2) 2-methyloxirane; (3) H30 Oc MCPBA OD (1) LIAIH4, ether: (2) H30* OE (1) CH3NH2, pH = 5: (2) NABH3CN O ) PPh3 (2) Buli: (3) benzaldehyde Oa(1) PPH3 (2) ĐuL: (3) acetophenone OK() Mg, ether; (2) acetone; (3) H30 OL NaCIO2, NaH2PO4 2-methyl-2-butene ot (1)(COC)2. DMSO; (2) NEt3 OK (CH2)2NH, pH - 5 OL H2NNH2, KOH, heat OM None of the other reaction conditions listed will produce this overall transformation ON CrO3, H2504 H20 o. H2NNH2, pH -5
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
From the list provided below, choose those observations that accurately describe the mineral presented in this image. A. This mineral does not have cleavage. B. This mineral effervesces with dilute...
-
Select one internal control from the list provided that would prevent or detect the error or fraud described. Controls may be used twice in this exercise or not at all. 1. ___ Invoices for goods sold...
-
The oscillation of rod OA about O is defined by the relation ( = 4/)(sin t), where and t are expressed in radians and seconds, respectively. Collar B slides along the rod so that its distance from O...
-
Pick a company or brand or business on which to focus. What business is it inP Who are its direct and indirect competitors? Which in each category are the most relevant competitors?
-
Why is it that some economists are suspicious that some of those who claim they are for "free trade but fair trade" are not really championing the cause of free trade?
-
State whether each of the following is an example of a physical or chemical property of matter: (a) Carbon dioxide escapes when a soda can is opened. (b) A bronze statue turns green. (c) A silver...
-
Water flows in a rectangular channel at a rate of \(q=20 \mathrm{cfs} / \mathrm{ft}\). When a Pitot tube is placed in the stream, water in the tube rises to a level of \(4.5 \mathrm{ft}\) above the...
-
Kit Chen has risen to the occasion and answered your questions concerning the policy for handling unfilled orders at Kit Chen Kaboodle, Inc. Based on those answers and any assumptions you need to...
-
Majesty Company uses target costing to ensure that its products are profitable. Assume Majesty is planning to introduc product with the following estimates: Estimated market price Annual demand Life...
-
Euro Corporation, a U.S. corporation, operates through a branch in Germany. During 2022, the branch reported taxable income of $1,000,000 and paid German income taxes of $300,000. In addition, Euro...
-
Companies that include both outside and inside salespeople often: Question 5 options: 1) compete for sales and customers 2) duplicate their efforts and increase the company's cost per sale 3) work...
-
a. What is the immediate change in the quantity of real GDP demanded? b. In the short run, does real GDP increase by more than, less than, or the same amount as the immediate change in the quantity...
-
Smith and Dudley are next-door neighbors. The branches of a large maple tree on Smiths property hangs over Dudleys driveway, dripping sap onto Dudleys car. Smith refuses to trim the branches...
-
Compute the reactions and draw the shear and moment curves for the beam in Figure P9.6. EI is constant. 15 kips B 15 kips 612126
-
What differences exist among the principal kinds of automobile insurance?
-
Suppose that Gen Z comes strongly out of the pandemic. Explain the effect of a rise in expenditure by Gen Z on real GDP and the price level in the short run. A 2018 study estimated Gen Zs spending...
-
According to Vygotsky's semiotic mediation theory, how do signs mediate human action?
-
Saccharin is an artificial sweetener that is used in diet beverages. In order for it to be metabolized by the body, it must pass into cells. Below are shown the two forms of saccharin. Saccharin has...
-
In the addition of HBr to 3,3-dimethyl-l-butene, the results observed are shown in Fig. P5.36. (a) Explain why the different conditions give different product distributions. (b) Write a detailed...
-
Which of the, two phenols in each set is more acidic? Explain. Phenol or m-chlorophenol
-
Give the products formed when each of the following pairs reacts in a Diels-Alder reaction; show the relative stereochemistry of the substituent groups where appropriate. Show both exo and endo...
-
What does accessibility mean?
-
Building trust in a company by an applicant for getting a job a. Explain the above scenario. b. Draw a class diagram for it. c. Document a detailed and significant use case. d. Create a sequence...
-
Building trust in parents by hostel authorities to state that hostel is safe for students to live a. Explain the above scenario. b. Draw a class diagram for it. c. Document a detailed and significant...

Study smarter with the SolutionInn App


