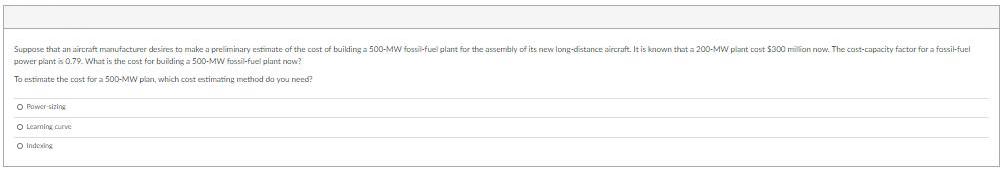
Suppose that an aircraft manufacturer desires to make a preliminary estimate of the cost of building...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
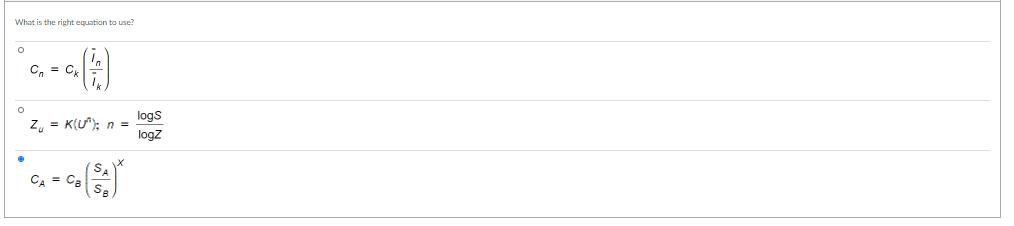
Suppose that an aircraft manufacturer desires to make a preliminary estimate of the cost of building a 500-MW fossil-fuel plant for the assembly of its new long-distance aircraft. It is known that a 200-MW plant cast $300 million now. The cast-capacity factor for a fassi-fuel power plant is 0.79. What is the cost for buiding a 500-MW fossil-fuel plant naw? To estimate the cast for a 500-MW plan, which cost estimating method da you need? O Power sizing O Leaming curve O Indexing What is the cost of the new 500-MW pian? O S300 milion O Ss00 millioe O $200 milion O $61872 milIfon Suppose that an aircraft manufacturer desires to make a preliminary estimate of the cost of building a 500-MW fossil-fuel plant for the assembly of its new long-distance aircraft. It is known that a 200-MW plant cast $300 million now. The cast-capacity factor for a fassi-fuel power plant is 0.79. What is the cost for buiding a 500-MW fossil-fuel plant naw? To estimate the cast for a 500-MW plan, which cost estimating method da you need? O Power sizing O Leaming curve O Indexing What is the cost of the new 500-MW pian? O S300 milion O Ss00 millioe O $200 milion O $61872 milIfon
Expert Answer:
Related Book For 

College Physics
ISBN: 978-0495113690
7th Edition
Authors: Raymond A. Serway, Jerry S. Faughn, Chris Vuille, Charles A. Bennett
Posted Date:
Students also viewed these mathematics questions
-
Make an order-of-magnitude estimate of the cost of one persons routine use of a hair dryer for 1 yr. If you do not use a blow dryer yourself, observe or interview someone who does. State the...
-
Make an order-of-magnitude estimate of the cost of one persons routine use of a hair dryer for 1 yr. If you do not use a blow dryer yourself, observe or interview someone who does. State the...
-
Refer to Exercise 79. Suppose a preliminary estimate of the population standard deviation will come from a pilot sample of five new accountants showing the following values: Determine the appropriate...
-
The Total Landed Costs (TLC) for a plastic wrap packaging machine from Germany to Monterrey, Mexico was calculated to be $723,566 USD. The company intends to use it for only 3 years and then resell...
-
What is the after-tax cost in 2013 of deductible interest expense of $9,000 and property taxes of $2,500 for a single taxpayer with gross income of $80,000? The taxpayer's only other itemized...
-
Are you in favor of the ruinous economic policy of the Democratic Platform Committee? Identify the fallacies of relevance, weak induction, presumption, ambiguity, and illicit transference committed...
-
The following information has been extracted from the financial statements and the notes of Champigon Ltd. Required (a) Calculate the following for 2023 to one decimal place: i. current ratio ii....
-
Crest Industries sells a single model of satellite radio receivers for use in the home. The radios have the following price and cost characteristics: Sales price . . . . . . . . . . . $ 80 per radio...
-
Let C' be the curve of intersection of the cylinder 25x + y = 25 and the plane x + y + z = 4. (a) Using t as the parameter where 0
-
During 2021, Brewster Company earned revenues of $146 million. Brewster incurred, during that same year, salary expense of $28 million, rent expense of $23 million, and utilities expense of $19...
-
Hoping to boost its revenues, a company is considering a credit policy extension, with the following summary taken into account: Add. ROI Additional sales Uncollectible accounts Collection costs...
-
You are a researcher gathering data to answer the following research question: Will improving preparation methods result in higher quiz grades? You decide to interview your fellow students and ask...
-
Show detailed mechanisms for the electrophilic addition of HCl with 2 - methyl - 1 , 3 - cyclohexadiene. Provide all possible adducts. Rank the order of most to least major products. Specify 1 , 2...
-
What role do social control mechanisms play in regulating deviant behavior, and how do these mechanisms vary across different cultural and historical contexts ?
-
You observe the spots prices and futures prices of KUY stock in the following table. Determine if the futures prices are accurately priced. Show you calculation. Assume interest rate is 4.8%p.a. and...
-
Suppose the exchange rate is $1.59/, the euro-denominated continuously compounded interest rate is 6%, the U.S. dollar-denominated continuously compounded interest rate is 5%, and the exchange rate...
-
Identify a manager who is highly concerned for production and uses rewards to maintain the loyalty of employees?
-
What are the two methods used to translate financial statements and how does the functional currency play a role in determining which method is used?
-
To repair a power supply for a stereo amplifier, an electronics technician needs a 100-F capacitor capable of withstanding a potential difference of 90 V between its plates. The only available supply...
-
A transmission line that has a resistance per unit length of 4.50 x 104 /m is to be used to transmit 5.00 MW over 400 miles (6.44 x 105 m). The output voltage of the generator is 4.50 kV (rms). (a)...
-
A ray of light strikes the midpoint of one face of an equiangular (606060) glass prism (n = 1.5) at an angle of incidence of 30. (a) Trace the path of the light ray through the glass, and find the...
-
A closed system passes from state 1 to state 2 while \(200 \mathrm{~kJ}\) of heat is added and \(280 \mathrm{~kJ}\) of work is done. As the system is returned to state \(1,160 \mathrm{~kJ}\) of work...
-
0.03 m 3 of air at a pressure of 1.2 bar and \(298 \mathrm{~K}\) is compressed to a volume of \(0.0045 \mathrm{~m}^{3}\) according to the law \(p V^{1.3}=\) const. Find the final temperature and work...
-
A system contains \(0.15 \mathrm{~m}^{3}\) of a gas at a pressure of 3.8 bar and \(423 \mathrm{~K}\). It is expanded adiabatically till the pressure falls to \(1 \mathrm{bar}\). The gas is then...

Study smarter with the SolutionInn App



