1. If 0.50 moles of C;H3 are allowed to react with 2.8 moles of O, to...
Fantastic news! We've Found the answer you've been seeking!
Question:


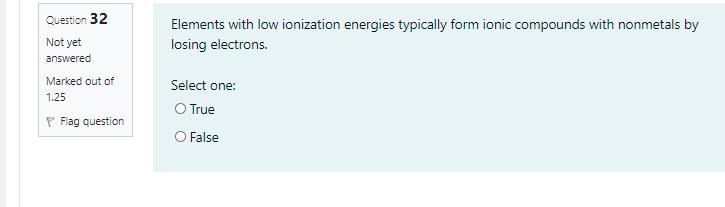
Transcribed Image Text:
1. If 0.50 moles of C;H3 are allowed to react with 2.8 moles of O, to produce carbon dioxide (CO,) and water (H,O), according to the following equation: 5 CO2 + 4 H20 CSH3 + 702 [Molar mass (g/mol) of C = 12.01, H = 1.008, and O = 16.00] Which of the following is the limiting reactant? а. О O b. H20 O. CO2 O d. CsH3 Question 30 All orbitals of a given subshell must be singly occupied before pairing begins in Not yet answered that subshell is a statement of Marked out of 1.25 P Flag question O a. Arrhenius definition O b. Pauli principle O c. the Aufbau Principle O d. Bronested concept O e. Hund's Rule Question 32 Elements with low ionization energies typically form ionic compounds with nonmetals by Not yet losing electrons. answered Marked out of Select one: 1.25 O True P Flag question OFalse 1. If 0.50 moles of C;H3 are allowed to react with 2.8 moles of O, to produce carbon dioxide (CO,) and water (H,O), according to the following equation: 5 CO2 + 4 H20 CSH3 + 702 [Molar mass (g/mol) of C = 12.01, H = 1.008, and O = 16.00] Which of the following is the limiting reactant? а. О O b. H20 O. CO2 O d. CsH3 Question 30 All orbitals of a given subshell must be singly occupied before pairing begins in Not yet answered that subshell is a statement of Marked out of 1.25 P Flag question O a. Arrhenius definition O b. Pauli principle O c. the Aufbau Principle O d. Bronested concept O e. Hund's Rule Question 32 Elements with low ionization energies typically form ionic compounds with nonmetals by Not yet losing electrons. answered Marked out of Select one: 1.25 O True P Flag question OFalse
Expert Answer:
Answer rating: 100% (QA)
31 Each mole of C5H8 would react with 7 moles of O2 w... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A series of dialkyl ethers was allowed to react with excess hydrogen bromide, with the following results. Identify the ether in each case. (a) Another ether gave only benzyl bromide. (b) A third...
-
(a) If excess C is allowed to react with 25.0 g of CO2 in a 3.00-L vessel at 1000 K, how many grams of CO are produced? (b) How many grams of C are consumed? (c) If a smaller vessel is used for the...
-
Aluminum reacts with chlorine gas according to the following equation shown below. How many moles of Cl 2 are required to react with 0.11 mol of Al? 2 Al( s ) + 3 Cl 2 ( g ) 2 AlCl 3 ( s )
-
In a law firm consisting of 20 lawyers, 9 are criminal lawyers, 6 are divorce lawyers, and 4 are both criminal and divorce lawyers. If a lawyer from this firm is chosen at random, what is the...
-
1. Do Bill and Molly qualify for any other tax-advantaged saving vehicles? If so, which ones? To what extent? 2. Since Bill does not receive an employer match, should he invest the maximum amount in...
-
Using the appropriate interest table, compute the present values of the following periodic amounts due at the end of the designated periods. (a) $30,000 receivable at the end of each period for 8...
-
Petitioner Salman was indicted for federal securities-fraud crimes for trading on inside information he received from a friend and relative-by-marriage, Michael Kara, who, in turn, had received the...
-
Weighted-average method. Ashworth Handcraft is a manufacturer of picture frames for large retailers. Every picture frame passes through two departments: the assembly department and the finishing...
-
B: Point out the error in the code and correct it. class Person { public void int display () { System.out.println("This is a Person"); } } class Student extnd Person { public void display () {...
-
Identify the compound with molecular formula C7H14O that gives the following proton-coupled 13C NMR spectrum. 80 60 40 20 o (ppm) frequency
-
Southern Fields has an inventory of 1,131,823 pounds of sugar. The firm placed a partial hedge on this inventory by selling 6 futures contracts at 9.39. The futures contracts are based on 112,000...
-
A farmer buys seed for 15 that is used to grow wheat. The farmer sells the wheat to themiller for 30 cents, who makes flour. The flour is sold to the baker for 60 cents and the bakermakes bread,...
-
Assume that you purchased a commercial (non-residential) property for $10,000,000 on May 31, 2018, with 20% of the purchase price attributed to land value. What is the total amount of depreciation...
-
Yesterday, a pairs of UGG boots cost $200, and Tom Brady was willing to buy 2 pairs. Today, UGG boots are on sale for $150, and Tom is now willing to buy 4 pairs. Is Toms demand for UGGs elastic or...
-
Just a simple question: Can there be a mixed strategy Nash equilibrium for only one player and not the other in a 2 x 2 matrix game?
-
The crate has now reached a ramp, that is inclined at an angle 15 with the horizontal. You push a crate up the ramp in a straight line, at a constant speed, over a distance d = 6 m along the ramp,...
-
The Indian authorities recently announced that they would allow foreign investors to buy Indian government bonds denominated in Rupees (INR) in the secondary market (i.e., the market where existing...
-
Which of the following streaming TV devices does not involve use of a remote controller? A) Google Chromecast B) Apple TV C) Amazon Fire TV D) Roku
-
Give the structure of the cycloaddition product formed when benzyne is generated in the presence of furan. (See Section 11.21, if necessary, to remind yourself of the structure of furan.)
-
31P is the only phosphorus isotope present at natural abundance and has a nuclear spin of 1/2The 1H NMR spectrum of trimethyl phosphite, (CH3O)3P, exhibits a doublet for the methyl protons with a...
-
(a) Which primary alcohol of molecular formula C5H12O cannot be prepared from an alkene? Why? (b) Write equations describing the preparation of three isomeric primary alcohols of molecular formula...
-
List all of the factors you can think of that people use when deciding where to shop for clothes.
-
List the most important three factors for you personally when deciding where to shop for clothes.
-
Intel has dominated the computer chip industry. Beginning with the iconic advertising campaign Intel Inside, the company has created a large market for powerful processors. This has allowed Intel to...

Study smarter with the SolutionInn App


