Question 4 Write the valence molecular orbital configuration of F2-. The fill order for F2- is...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
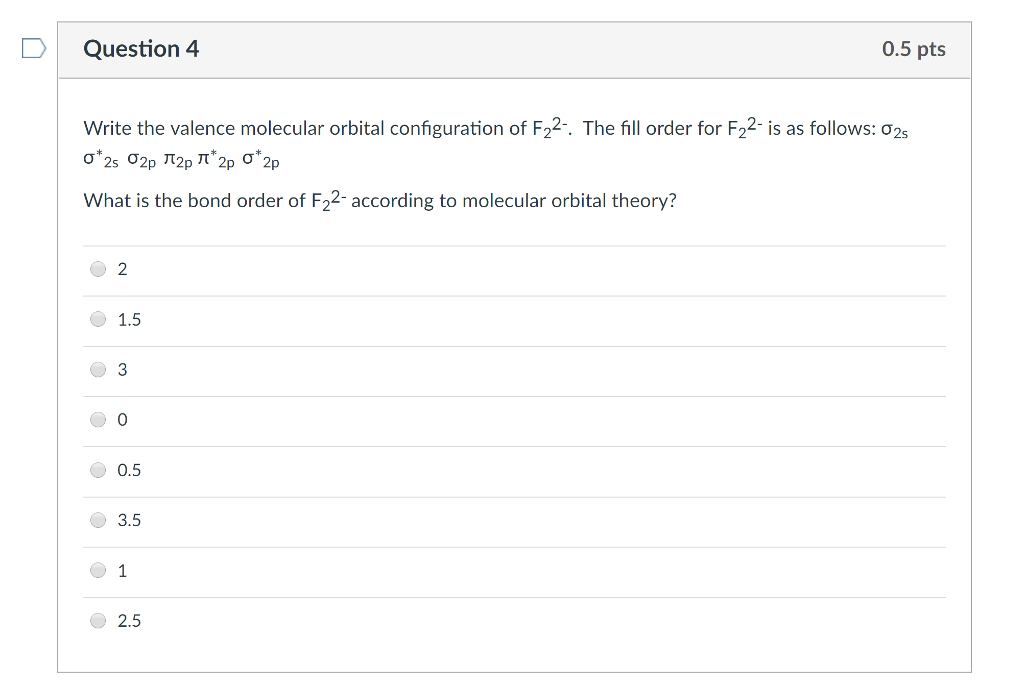
Question 4 Write the valence molecular orbital configuration of F₂2-. The fill order for F₂2- is as follows: 02s 0*2s 2p ¹p ¹*2p O*2p What is the bond order of F₂2- according to molecular orbital theory? O O 2 1.5 3 0 0.5 3.5 1 0.5 pts 2.5 Question 4 Write the valence molecular orbital configuration of F₂2-. The fill order for F₂2- is as follows: 02s 0*2s 2p ¹p ¹*2p O*2p What is the bond order of F₂2- according to molecular orbital theory? O O 2 1.5 3 0 0.5 3.5 1 0.5 pts 2.5
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
A condensed income statement for Inman Company is as follows for the month of November: Further analysis revealed the following data on costs: Required: a. Prepare a report comparing the master...
-
The orbital diagram that follows shows the valence electrons for a 2+ ion of an element. (a) What is the element? (b) What is the electron configuration of an atom of this element? 4d
-
Write the molecular orbital configuration of the diatomic molecule BN. What is the bond order of BN? Is the substance diamagnetic or paramagnetic? Use the order of energies that was given for...
-
A. Discuss the following conceptual model/framework of Orem's Self-Care Theory CONDITIONING FACTORS (12 MARKS) Orem's Self-Care Theory Conceptual Framework SELF-CARE AGENCY SELF-CARE DEFICIT NURSING...
-
Major League Apparel has two classes of stock authorized: 6%, $10 par preferred, and $1 par value common. The following transactions affect stockholders equity during 2015, its first year of...
-
Financial statement data for years ending December 31 for Chiro-Solutions Company are shown below. a. Determine the accounts receivable turnover for 2014 and 2013. b. Determine the number of days'...
-
On March 13, 1963, Ernesto Miranda was arrested for kidnapping and rape and taken to a Phoenix police station. After being identified by the complaining witness, he was questioned by two police...
-
Each of the following items must be considered in preparing a statement of cash flows (indirect method) for Turbulent Indigo Inc. for the year ended December 31, 2014. a) Plant assets that had cost...
-
Find the solution of the equation coth x = a, assuming that a > 1. Express the result in terms of elementary functions of a. [Hint: The result should have the form x = f(a), where f(a) is a specific...
-
Fawcett Institute provides one-on-one training to individuals who pay tuition directly to the business and also offers extension training to groups in off-site locations. Fawcett prepares adjusting...
-
Consider two charged particles, one with +2 C and one with -5 C of charge. For each one, think about what the electric field lines around the charge would look like. 1. Describe in a few sentences...
-
A rod of length 38.50 cm has linear density (mass per length) given by = 50.0 + 18.0x where x is the distance from one end, and 1 is measured in grams/meter. (a) What is its mass? g (b) How far from...
-
In what ways do power structures and systems of privilege and oppression influence the trajectory of social change movements, and how can intersectional approaches address the complexities of...
-
Explain what the "so-called corporate veil" is and what is meant by lifting the veil in common law? B) Critically discuss the circumstances when the corporate veil can be lifted by the court.
-
The corporate veil is an established principle in company law. There circumstances when the veil can be lifted. Discuss when the veil can be kifted in about 400 words ?
-
An established medium sized resort in Spain is known for its unspoiled beaches and gently rolling acres bordering a national park. The resort has an excellent reputation with an international...
-
Two firms, A and B, both produce widgets. The price of widgets is $1 each. Firm A has total fixed costs of $500,000 and variable costs of 50 per widget. Firm B has total fixed costs of $240,000 and...
-
H.J. Heinzs annual dividends were as follows: 1990 ..............$0.540 1991.............. 0.620 1992 .............. 0.700 1993.............. 0.780 1994 .............. 0.860 1995 .............. 0.940...
-
One or both of the following have geometric isomers. Draw the structures of any geometric isomers. a. b. CH:CHCH-CHCH3 CH3 CH3
-
What is meant by the reference form of an element? What is the standard enthalpy of formation of an element in its reference form?
-
(a) Formaldehyde, H2CO, is a colorless, pungent gas used to make plastics. Give the valence bond description of the formaldehyde molecule. (Both hydrogen atoms are attached to the carbon atom.) (b)...
-
At January 1, 2015, Cheng Company reported retained earnings of 20,000,000. In 2015, Cheng discovered that 2014 depreciation expense was understated by 4,000,000. In 2015, net income was 9,000,000...
-
Cherokee Construction Company changed from the cost-recovery to the percentage-of-completion method of accounting for long-term construction contracts during 2015. For tax purposes, the company...
-
In 2015, Bailey Corporation discovered that equipment purchased on January 1, 2013, for 50,000 was expensed at that time. The equipment should have been depreciated over 5 years, with no residual...

Study smarter with the SolutionInn App



