How many grams of lithium nitrate will be needed to make 200.0 grams of lithium sulfate,...
Fantastic news! We've Found the answer you've been seeking!
Question:
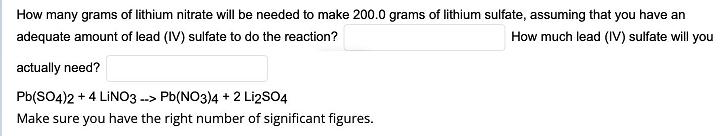
Transcribed Image Text:
How many grams of lithium nitrate will be needed to make 200.0 grams of lithium sulfate, assuming that you have an adequate amount of lead (IV) sulfate to do the reaction? How much lead (IV) sulfate will you actually need? Pb(SO4)2 + 4 LİNO3 --> Pb(NO3)4 + 2 Li2SO4 Make sure you have the right number of significant figures. How many grams of lithium nitrate will be needed to make 200.0 grams of lithium sulfate, assuming that you have an adequate amount of lead (IV) sulfate to do the reaction? How much lead (IV) sulfate will you actually need? Pb(SO4)2 + 4 LİNO3 --> Pb(NO3)4 + 2 Li2SO4 Make sure you have the right number of significant figures.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
How many grams of perchloric acid, HClO4, are contained in 37.6 g of 70.5 wt% aqueous perchloric acid? How many grams of water are in the same solution?
-
How many grams of nitrogen, N, are there in a fertilizer containing 48.5 g of ammonium nitrate and no other nitrogen-containing compound? See Exercise 3.7 for the percentage composition of NH4NO3.
-
How many grams of oxygen gas are required to produce 7.60 kJ of heat when hydrogen gas burns at constant pressure to produce gaseous water? 2H2(g) + O2(g) 2H2O(g); H = 484 kJ Liquid water has a heat...
-
CPA firm brings in a yoga instructor during the tax busy season to help relieve stress of the employees. Which is true about the CPA firm's ability to take a deduction for the yoga instructor's...
-
Shown below are three containers of an ideal gas (A, B, and C), each equipped with a movable piston (assume that atmospheric pressure is 1.0 atm). a. How do the pressures in these containers compare?...
-
Refer to the Arctic Springs Bottling Department Data Set. 1. Draw a time line. 2. Complete the first two steps of the process costing procedure for the Bottling Department: summarize the physical...
-
Suppose that we wish to know the standard deviation of the phase of \(\mathbf{J}_{12}(T)\) under the condition that the measurement time \(T\) is sufficiently long compared with the correlation time...
-
One of the approaches for the Southern Company to comply with the Clean Air Act is to adopt fuel-switching technology. Do you think this strategic flexibility would have value to Southern Companys...
-
Income Statement Calculations O'Connor Company's income statement information is as follows: Year 2 Year 3 Beginning inventory $ (a) $ (d) Sales 206,000 (e) Purchases 130,000 140,000 Purchases...
-
Using Pubs (or Pubs2) sample tables to create SQL statements (1 per question) to accomplish the following (command and output required): List the first name, last name and author ID of the authors...
-
A client has given you several units to lease in the same block. The lowest priced one ($350/week) has been successfully leased but the landlord doesn't want a new signboard with the new price on it...
-
For power-system fault studies, it is assumed that the system is operating under balanced steady-state conditions prior to the fault, and sequence networks are uncoupled before the fault occurs. (a)...
-
Suppose that last month the U.S. dollar was trading on the foreign-exchange market at 0.85 euro per dollar, and today the U.S. dollar is trading at 0.88 euro per dollar. Explain what has hap pened....
-
Which statement about oligopolies is false? a) They operate at the minimum points of their ATC curves. b) They charge higher prices than perfect competitors. c) They make profits in the long run. d)...
-
Practical Application: Suppose you just purchased a mens clothing store in a huge shop ping mall. How would you differentiate your stores wares from those of your competitors?
-
The entire flow of U.S. dollars and foreign currencies into and out of the country constitutes our ________.
-
If a Department Manager is hired to strictly directly assist the clinic in its routine operations and making $6,000 per month, is that a direct expense?
-
(a) Prove that form an orthonormal basis for R3 for the usual dot product. (b) Find the coordinates of v = (1, 1, 1)T relative to this basis. (c) Verify formula (5.5) in this particular case. 48-65...
-
Describe how you could separate the following mixture of metal ions: Na+, Hg2+, and Ca2+.
-
Promethium-147 has been used in luminous paint for dials. The half-life of this isotope is 2.5 y. What is the decay constant (in /s)?
-
Hydrogen sulfide is a gas with the odor of rotten eggs. The gas can sometimes be detected in automobile exhaust. It is a compound of hydrogen and sulfur in the atomic ratio 2 : 1. A sample of...
-
Explain how the financial manager might use industry norms in the design of the companys financing mix.
-
You have developed the following income statement for Sing-Tel Corporation. It represents the most recent years operations, which ended yesterday. Your supervisor in the controllers office has just...
-
Footwear, Inc. manufactures a complete line of mens and womens formal shoes for independent merchants. The average selling price of its finished product is \($85\) per pair. The variable cost for...

Study smarter with the SolutionInn App



