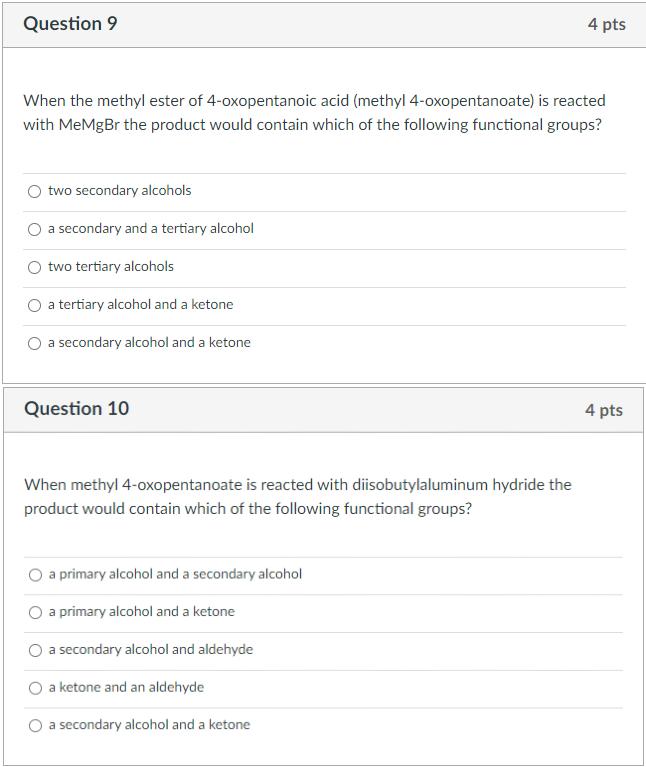
Question 9 4 pts When the methyl ester of 4-oxopentanoic acid (methyl 4-oxopentanoate) is reacted with...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Question 9 4 pts When the methyl ester of 4-oxopentanoic acid (methyl 4-oxopentanoate) is reacted with MeMgBr the product would contain which of the following functional groups? two secondary alcohols a secondary and a tertiary alcohol two tertiary alcohols a tertiary alcohol and a ketone a secondary alcohol and a ketone Question 10 4 pts When methyl 4-oxopentanoate is reacted with diisobutylaluminum hydride the product would contain which of the following functional groups? a primary alcohol and a secondary alcohol a primary alcohol and a ketone a secondary alcohol and aldehyde a ketone and an aldehyde a secondary alcohol and a ketone Question 9 4 pts When the methyl ester of 4-oxopentanoic acid (methyl 4-oxopentanoate) is reacted with MeMgBr the product would contain which of the following functional groups? two secondary alcohols a secondary and a tertiary alcohol two tertiary alcohols a tertiary alcohol and a ketone a secondary alcohol and a ketone Question 10 4 pts When methyl 4-oxopentanoate is reacted with diisobutylaluminum hydride the product would contain which of the following functional groups? a primary alcohol and a secondary alcohol a primary alcohol and a ketone a secondary alcohol and aldehyde a ketone and an aldehyde a secondary alcohol and a ketone
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
When an aldehyde or a ketone is condensed with ethyl a-chloroacetate in the presence of sodium ethoxide, the product is an α,β-epoxy ester called a glycidic ester. The...
-
Which of the following functional groups will appear in the range 160-220 ppm in 13C NMR? A) ethoxy group B) aromatic carbons C) carbonyl group D) methoxy group
-
An automobile engine uses methyl alcohol (CH3OH) as fuel with 200 percent excess air. Air enters this engine at 1 atm and 25oC. Liquid fuel at 25oC is mixed with this air before combustion. The...
-
Below are three independent and unrelated errors. a. On December 31, 2020, Wolfe-Bache Corporation failed to accrue salaries expense of $1,800. In January 2021, when it paid employees for the...
-
Refer to the data of Exercise 11.8. a. Calculate a 95% confidence interval for 1. b. What is the interpretation of H0: 1 = 0 in Exercise 11.8? c. What is the natural research hypothesis Ha for the...
-
In an economy, the natural unemployment rate is 4 percent and the expected inflation rate is 3 percent a year. Draw a graph of the short-run and long-run Phillips curves that display this...
-
There are 24,627 species of fish on Earth. Decide whether the statement makes sense (or is clearly true) or does not make sense (or is clearly false). Explain clearly.
-
In exercise 12, the following data on x = average daily hotel room rate and y = amount spent on entertainment (The Waal Street Journal, August 18, 2011) led to the estimated regression equation y =...
-
William works in a factory sorting packages on a roller - belt conveyor system. Feeling a bit impatient, he helps move a 1 4 . 6 - kg package along by pushing it over a distance of 3 . 2 m with a...
-
Why have many institutions moved away from the dormitory concept to smaller rooms occupied by two or three juveniles? allows for separation between types of people who are incarcerated makes the...
-
Part A In the accompanying figure, the chance that individual IV-7 is a heterozygous carrier is | || ||| IV V 00000 3/4 1/4 2/3 1/2 1/3 18 8 do 10 10 """b's 13 14 13 11 12 15 HO's 114 15
-
Write a short essay (500 words) listing the advantages and disadvantages of having the same accounting rules in all countries.
-
Explain the following: petroleum reservoir primary recovery secondary recovery tertiary recovery
-
Write down the documentation you would expect to see as evidence of the money amount of the following liabilities: bank overdraft; amount owing to a trade supplier
-
Define the following terms: day-rate contract footage-rate contract turnkey contract horizontal drilling
-
A news alert contains the following statement: Telecoms plc this week raised cash by selling $50m bonds with five-year and ten year maturities. Explain each part of the sentence.
-
A cooperative allocated $5,000 of total allocated equity to patrons for the 2022 fiscal year, $3,000 total allocated equity to patrons for the 2023 fiscal year, and $6,000 total allocated equity to...
-
a) Calculate the goodwill that was paid by Major Ltd on the acquisition of Minor Ltd. [10 marks] b) Prepare the consolidated statement of financial position for Major Ltd at 31 July 20X8. [30 marks]...
-
Would RNA of a certain percentage composition of guanine and cytosine denature more or less readily than RNA with a lower percent of guanine and cytosine? Why?
-
Ozone decomposes to oxygen gas. 2O3(g) 3O2(g) A proposed mechanism for this decomposition is What is the rate law derived from this mechanism? O3 2 + O (fast, equilibrium) slow)
-
Consider the voltaic cell Cd(s) |Cd2+(aq) || Ni2+(aq) | Ni(s) Write the half-cell reactions and the overall cell reaction. Make a sketch of this cell and label it. Include labels showing the anode,...
-
Which of the following mechanisms of evolution consistently causes populations to become more adapted to their environments? (a) natural selection (b)mutation pressure (c) genetic drift (d) gene flow
-
Which of the following provides evidence for evolution? (a) changes in the coloration of peppered moth populations over time (b) the presence of vestigial eyes in cave salamanders (c) the fact that...
-
Photosynthesizing plants are (a) heterotrophs. (b) autotrophs. (c) chemoautotrophs. (d) archaeans.

Study smarter with the SolutionInn App


