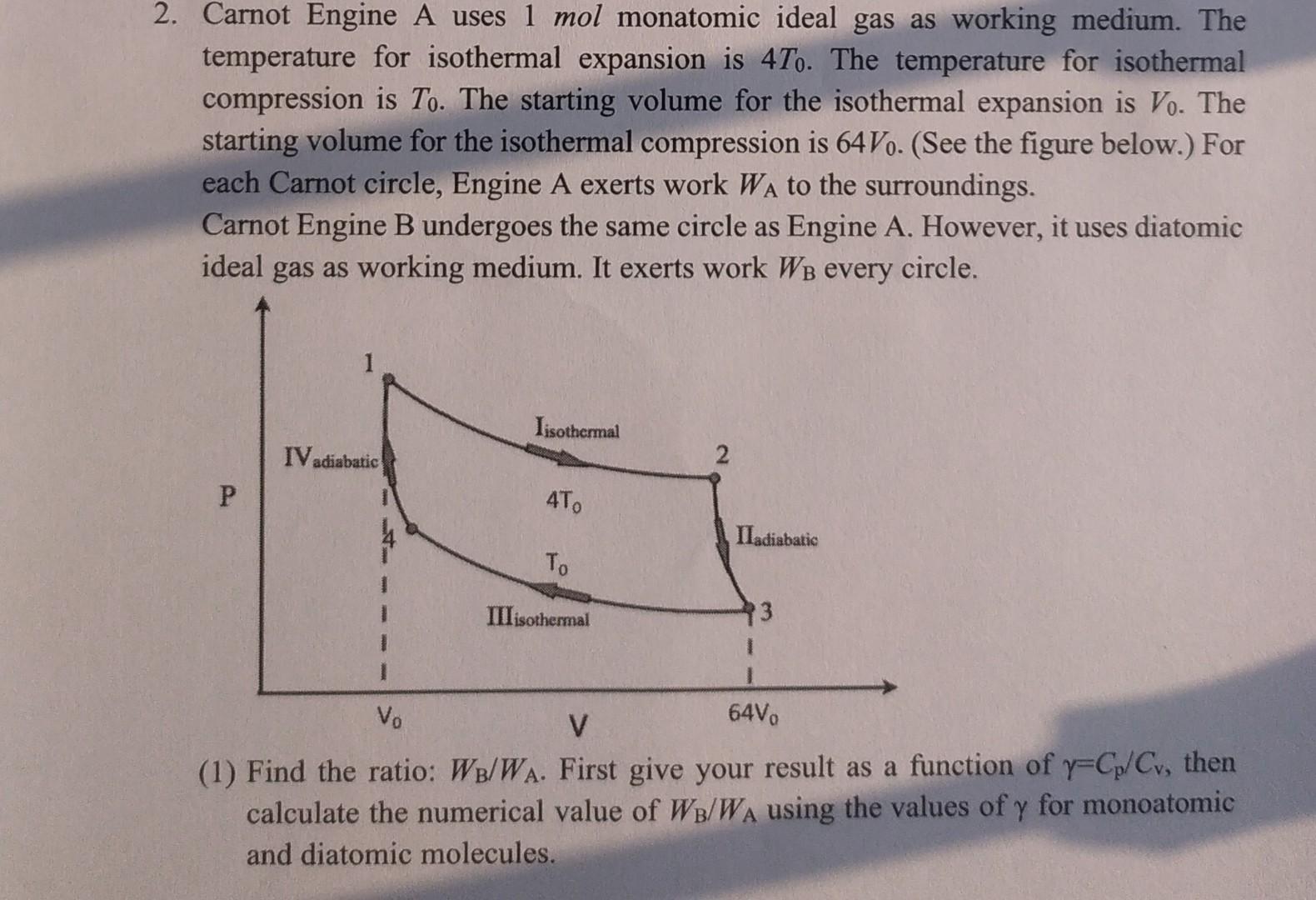
2. Carnot Engine A uses 1 mol monatomic ideal gas as working medium. The temperature for...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
2. Carnot Engine A uses 1 mol monatomic ideal gas as working medium. The temperature for isothermal expansion is 47o. The temperature for isothermal compression is To. The starting volume for the isothermal expansion is Vo. The starting volume for the isothermal compression is 64Vo. (See the figure below.) For each Carnot circle, Engine A exerts work WA to the surroundings. Carnot Engine B undergoes the same circle as Engine A. However, it uses diatomic ideal gas as working medium. It exerts work Wв every circle. P IV adiabatic Iisothermal 4To To III isothermal 2 IIadiabatic 13 Vo 64Vo V (1) Find the ratio: WB/WA. First give your result as a function of y-Cp/Cv, then calculate the numerical value of WB/WA using the values of y for monoatomic and diatomic molecules. 2. Carnot Engine A uses 1 mol monatomic ideal gas as working medium. The temperature for isothermal expansion is 47o. The temperature for isothermal compression is To. The starting volume for the isothermal expansion is Vo. The starting volume for the isothermal compression is 64Vo. (See the figure below.) For each Carnot circle, Engine A exerts work WA to the surroundings. Carnot Engine B undergoes the same circle as Engine A. However, it uses diatomic ideal gas as working medium. It exerts work Wв every circle. P IV adiabatic Iisothermal 4To To III isothermal 2 IIadiabatic 13 Vo 64Vo V (1) Find the ratio: WB/WA. First give your result as a function of y-Cp/Cv, then calculate the numerical value of WB/WA using the values of y for monoatomic and diatomic molecules.
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
In general, ionization energy increases from left to right across a given period. Aluminum, however, has a lower ionization energy than magnesium. Explain.
-
A space traveler is moving from left to right along the curve y = x2. When she shuts off the engine, she will continue traveling along the tangent line at the point where she is at that time. At what...
-
Calculate the ratio of the electrical and gravitational forces between a proton and an electron. Is neglect of the gravitational force justified?
-
Solve the problem. Jake Dowell has total fixed monthly expenses of $1367 and his gross monthly income is $3861. What is his debt-to-income ratio? Round to the nearest percent. ? 3% o 4% o 28% o 35%
-
Find each missing quantity using D N = d n. 1500 1. 18.0 2. 36.0 3. 12.0 9.00 6.00 9.00 972 1800 2250 1860 1125 620 5. 49.0
-
Which of the following statements, if added here, would best conclude the essay? F. NO CHANGE G. Now is when H. When J. OMIT the underlined portion.
-
Keurig Green Mountain is a specialty coffee company. It sources, makes, and sells coffee, hot cocoa, teas, and other beverages under various brands in portion packs for its Keurig brewing system. The...
-
Construct a graph showing equilibrium in the market for movie tickets. Label both axes and denote the initial equilibrium price and quantity as P0 and Q0. For each of the following events, draw an...
-
calculate both intrinsic value and time value for the options that you have picked. ANZ GROUP HOLDINGS LIMITED ANZ LAST PRICE / TODAY'S CHANGE $25.280+$0.350 (1.403 %) Industry Group: Banks VOLUME...
-
Consider the data below that shows the forecast for the number of cars sold by an electric car maker for each quarter for the next 5 years. The table below also shows the parameters for the...
-
A bank charges Iron Inc. an interest rate of 5.5% compounded quarterly on its line of credit. If 0.5% was added to the effective interest rate, calculate the new interest rate compounded quarterly....
-
List the various administrative sources of tax law.
-
Which of the types of statutory reorganization are "acquisitive"?
-
Explain what is meant by the terms "refundable" and "nonrefundable" credits. How might these classifications affect the tax liability of a taxpayer?
-
What potential advantage is there for a net overall capital gain situation?
-
Will a partnership under state law be taxed as a partnership under the Internal Revenue Code?
-
The current market price for common shares of Funny Company is $15. Put options on these shares currently trade at 0.50 and come with a $10 strike price. If the stock's market price falls to $8 what...
-
Match the following. Answers may be used more than once: Measurement Method A. Amortized cost B. Equity method C. Acquisition method and consolidation D. Fair value method Reporting Method 1. Less...
-
When 2-bromo-2,3-dimethylbutane reacts with a base under E2 conditions, two alkenes (2,3-dimethyl-1-butene and 2,3-dimethyl- 2-butene) are formed. a. Which of the bases shown would give the highest...
-
If 2-fluoropentane were to undergo an E1 reaction, would you expect the major product to be the one predicted by Zaitsev's rule? Explain.
-
a. Which of the following compounds will form hydrogen bonds between its molecules? 1. CH 3 CH 2 CH 2 COOH 2. CH 2 CH 2 N(CH 3 ) 2 3. CH 3 CH 2 CH 2 CH 2 Br 4. CH 3 CH 2 CH 2 NHCH 3 5. CH 3 CH 2 OCH...
-
Eugene Fama and Robert Shiller recently won the Nobel Prize in economics. Go to http://nobelprize.org/nobel_prizes/economics/ and locate the press release on Eugene Fama and Robert Shiller. What was...
-
Visit the Bloomberg Markets website at www.bloomberg.com/markets/stocks. Their interactive graph allows you to see cumulative returns for individual stocks as well as market indices. Over the last...
-
Compute the price of a share of stock that pays a \($5\) per year dividend and that you expect to be able to sell in one year for \($40\), assuming you require a 5% return.

Study smarter with the SolutionInn App


