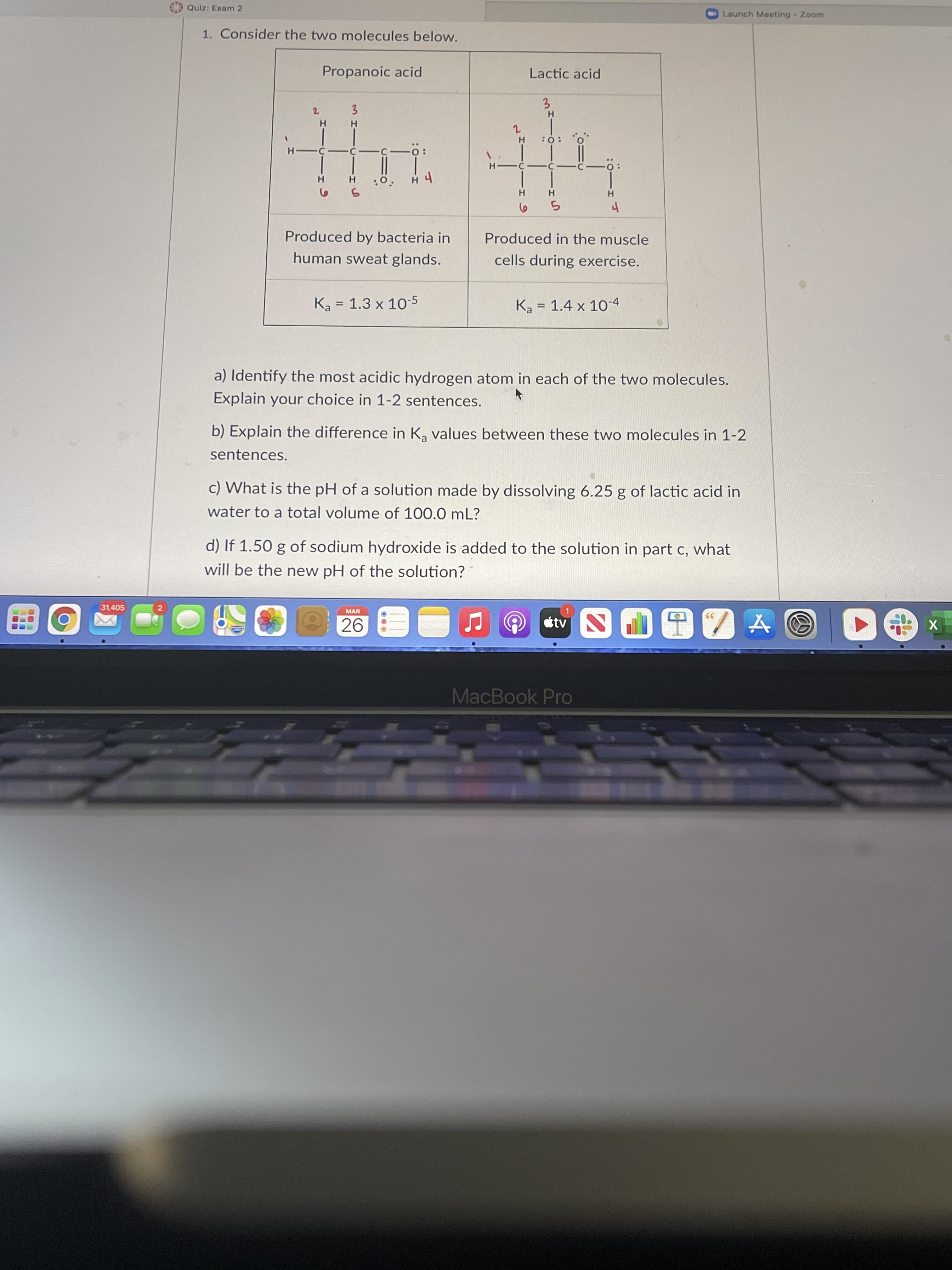
31,405 Quiz: Exam 2 1. Consider the two molecules below. Propanoic acid 2 3 H H...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
31,405 Quiz: Exam 2 1. Consider the two molecules below. Propanoic acid 2 3 H H H 2 HO: O H -c-c-c-Ö: -C- H :0. H 4 5 6 5 4 Produced by bacteria in human sweat glands. Produced in the muscle cells during exercise. Ka 1.3 x 10-5 = Ka 1.4 x 10-4 = a) Identify the most acidic hydrogen atom in each of the two molecules. Explain your choice in 1-2 sentences. b) Explain the difference in Ką values between these two molecules in 1-2 sentences. c) What is the pH of a solution made by dissolving 6.25 g of lactic acid in water to a total volume of 100.0 mL? d) If 1.50 g of sodium hydroxide is added to the solution in part c, what will be the new pH of the solution? 2 MAR 26 tv DIZA MacBook Pro H- Lactic acid 3 -Ö: H Launch Meeting - Zoom X 31,405 Quiz: Exam 2 1. Consider the two molecules below. Propanoic acid 2 3 H H H 2 HO: O H -c-c-c-Ö: -C- H :0. H 4 5 6 5 4 Produced by bacteria in human sweat glands. Produced in the muscle cells during exercise. Ka 1.3 x 10-5 = Ka 1.4 x 10-4 = a) Identify the most acidic hydrogen atom in each of the two molecules. Explain your choice in 1-2 sentences. b) Explain the difference in Ką values between these two molecules in 1-2 sentences. c) What is the pH of a solution made by dissolving 6.25 g of lactic acid in water to a total volume of 100.0 mL? d) If 1.50 g of sodium hydroxide is added to the solution in part c, what will be the new pH of the solution? 2 MAR 26 tv DIZA MacBook Pro H- Lactic acid 3 -Ö: H Launch Meeting - Zoom X
Expert Answer:
Answer rating: 100% (QA)
1a The proton attached with the carboxylic acid group is the most acidic one because of the equivale... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the pH of a solution in which 15 mL of 0.10 M NaOH has been added to 25 mL of 0.10 M HCl?
-
What is the pH of a solution in which 15 mL of 0.10 M NaOH is added to 25 mL of 0.10 M HCl?
-
What is the pH of a solution in which 40 mL of 0.10 M NaOH is added to 25 mL of 0.10 M HCl?
-
How do researchers investigate the effectiveness of the integrated reporting initiative?
-
Suppose that Country A and Country B both produce wine and cheese. Country A has 800 units of available labor, while Country B has 600 units. Prior to trade, Country A consumes 40 pounds of cheese...
-
A baseball pitcher throws a baseball horizontally at a linear speed of 42.5 m/s (about 95 mi/h). Before being caught, the baseball travels a horizontal distance of 16.5 m and rotates through an angle...
-
Which of the following is an activity not usually associated with forensic accounting and fraud examination consulting and litigation support? 1. A. Assessing fraud risk associated with internal...
-
Variable and absorption costing and breakeven points Mega-Air, Inc., manufactures a specialized snowboard made for the advanced snowboarder. Mega-Air began 2011 with an inventory of 240 snowboards....
-
A. Calculate the work, w , (in J) when 8.9 litre of an ideal gas at an initial pressure of 83.1 atm is expanded to a final pressure of 0.66 atm against a constant exteral pressure of 0.66 atm. Assume...
-
Toronto Cricket Property Inc. (TPI) has been incorporated with the purpose of build and operate a world-class cricket stadium outside Toronto. The land on which the stadium will be build is currently...
-
Attached is a series of errors made by Drew, an enthusiastic, but busy algebra student who did his practice test quickly and made some classic errors. In your group, talk about the many errors Drew...
-
Briefly summarize the article, and apply economic concepts to it. This is to include graphs, proper economic terminology, application of economic theory to the specifics of the article, and finally...
-
Which consumer insights can help inform Glossier's media plan? Explain. In other words, what should Glossier know about its target audience's media habits?
-
At a height of 3,500 m above sea level, the gage pressure in a car tire is measured using a pressure gage to be 0.25 MPa (1 megapascal=1 MPa = 1106 Pa). What is the absolute pressure in the car tire?...
-
David receives $3 a month as an allowance to spend as he pleases. Since he only likes peanut butter and jelly sandwiches, he spends the entire amount on peanut butter (at $0.05 per ounce) and jelly...
-
The video for BoA shows that the closing cost is $5619.41, i.e., a borrower will receive net $94380.59 (100K-5619) instead of borrowing 100,000. However, the monthly loan installments are the quoted...
-
1. Command to verify 'auditd' is active: [Enter answer here] 2. Command to set number of retained logs and maximum log file size: [Enter answer here] Add the edits made to the configuration file:...
-
Fill in each blank so that the resulting statement is true. 83 + 103 = ______ .
-
Let H k (n) be the number of vectors x 1 , . . ., x k for which each x i is a positive integer satisfying 1 x i n and x 1 x 2 . . . x k . (a) Without any computations, argue that H 1 (n) = n How...
-
An urn initially contains one red and one blue ball. At each stage, a ball is randomly chosen and then replaced along with another of the same color. Let X denote the selection number of the first...
-
You have $1000, and a certain commodity presently sells for $2 per ounce. Suppose that after one week the commodity will sell for either $1 or $4 an ounce, with these two possibilities being equally...
-
How does each of the following variables behave over the business cycle? Develop graphs to show your results and give economic explanations. a. Real imports b. Federal government receipts c. Housing...
-
In the FRED database, find a variable that is available in both a seasonally adjusted form and a not seasonally adjusted form. Plot both over time and describe how large the seasonal variation in the...
-
It has been argued that the stock market predicts recessions. Using quarterly data since 1961, plot the real value of the stock market index (the Wilshire 5000 index in the last month of the quarter...

Study smarter with the SolutionInn App


