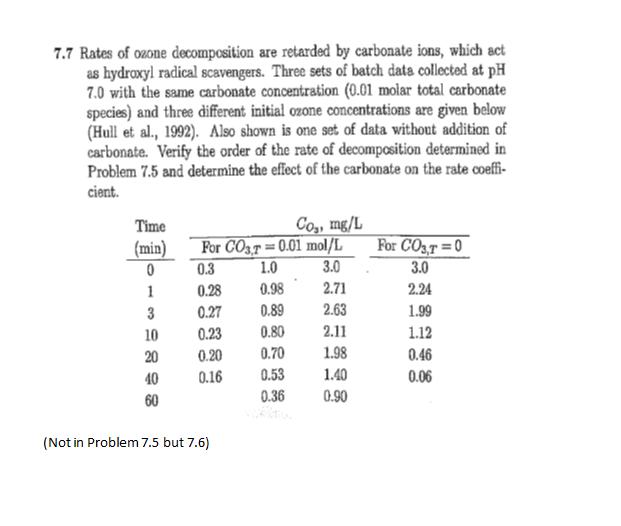
7.7 Rates of ozone decomposition are retarded by carbonate ions, which act as hydroxyl radical scavengers....
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
7.7 Rates of ozone decomposition are retarded by carbonate ions, which act as hydroxyl radical scavengers. Three sets of batch data collected at pH 7.0 with the same carbonate concentration (0.01 molar total carbonate species) and three different initial ozone concentrations are given below (Hull et al., 1992). Also shown is one set of data without addition of carbonate. Verify the order of the rate of decomposition determined in Problem 7.5 and determine the effect of the carbonate on the rate coeffi- cient. Time (min) 0 1 3 10 20 40 60 For CO3,7= 0.01 mol/L 0.3 1.0 3.0 0.98 2.71 0.89 2.63 0.80 2.11 0.70 1.98 0.53 1.40 0.36 0.90 0.28 0.27 0.23 0.20 0.16 Co₂, mg/L (Not in Problem 7.5 but 7.6) . For CO3,T=0 3.0 2.24 1.99 1.12 0.46 0.06 7.7 Rates of ozone decomposition are retarded by carbonate ions, which act as hydroxyl radical scavengers. Three sets of batch data collected at pH 7.0 with the same carbonate concentration (0.01 molar total carbonate species) and three different initial ozone concentrations are given below (Hull et al., 1992). Also shown is one set of data without addition of carbonate. Verify the order of the rate of decomposition determined in Problem 7.5 and determine the effect of the carbonate on the rate coeffi- cient. Time (min) 0 1 3 10 20 40 60 For CO3,7= 0.01 mol/L 0.3 1.0 3.0 0.98 2.71 0.89 2.63 0.80 2.11 0.70 1.98 0.53 1.40 0.36 0.90 0.28 0.27 0.23 0.20 0.16 Co₂, mg/L (Not in Problem 7.5 but 7.6) . For CO3,T=0 3.0 2.24 1.99 1.12 0.46 0.06
Expert Answer:
Answer rating: 100% (QA)
Solution Plotting each group of rate data as 1reaction time versus In03 wil... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Set out below are ratios relating to three different businesses. Each business operates within a different industrial sector. Required: State, with reasons, which of the above is: (a) A holiday tour...
-
Below are shown three different crystallographic planes for a unit cell of some hypothetical metal. The circles represent atoms: (a) To what crystal system does the unit cell belong? (b) What would...
-
Below you will find three data sets that have already been sorted. Fill in the blanks to find the upper and lower quartiles. Measurements Above and Below and Measurements Above and Below and Position...
-
What is the difference between average product and marginal product? Can you sketch a total product function such that the average and marginal product functions coincide with each other?
-
Since an absorbing gas re-emits the light it absorbs, why are there dark lines in an absorption spectrum? That is, why doesn't the re-emitted light simply fill in the dark places?
-
A bedroom bureau with a mass of 45 kg, including drawers and clothing, rests on the floor. (a) If the coefficient of static friction between the bureau and the floor is 0.45, what is the magnitude of...
-
How does encapsulation protect the object from an intruder?
-
A tank full of water has the shape of a paraboloid of revolution as shown in the figure; that is, its shape is obtained by rotating a parabola about a vertical axis. (a) If its height is 4 ft and the...
-
Estimate the value of using Simpson's rule. 15 x+1 15 dx by using n=4 subintervals of equal width
-
Coffee Blending and Sales Hill-O-Beans Coffee Company blends four component beans into three final blends of coffee: one is sold to luxury hotels, another to restaurants, and the third to...
-
Question 19 Which of the following is an example of a hydrometallurgical process? Al2O3.nH2O(s) + 2OH(aq) + 2 H2O(1) 2 Al(OH)4 (aq) CuSO4.5H2O(s) + H2O(1) CuS(s) + H2SO4(aq) +5 OH(aq) Al(OH)3(s) +...
-
Imagine that you pay $265 for a 4-year, 4% coupon bond with par value of $265.You intend to hold the bond for 1 year.That is, you are going to buy a 4-year bond and then 1 year later, you'll sell a...
-
If Adalyn Amusements Inc. has a 8-for-5 stock split and Jane owns 100 shares that were worth $123 per share before the split, what is one share worth immediately after the split?
-
In South Africa the national lottery is run weekly. In this lottery the purchaser of a ticket selects six different numbers from 1 to 50 inclusive. If those same six numbers are then drawn randomly...
-
Suppose you discovered a systematic relationship between the price earnings ratios of stocks and the performance of stocks. In other words, knowledge of a firm's price-earnings ratio proved helpful...
-
A firm contemplates buying an asset for $75,000.It will use the asset for 3 years, then sell it for $33,000.The sale will close out the asset's CCA (capital cost allowance) account.The CCA rate for...
-
The body mass index, or BMI, is a calculation used to determine your level of body fat. BMI is a simple calculation using a person's height and weight. In some cases, it can help a doctor determine...
-
Ask students to outline the reasons why the various elements of culture (social structures and control systems, language and aesthetics, religion and other belief systems, educational systems, etc.)...
-
The designations 1A through 8A used for certain families of the periodic table are helpful for predicting the charges on ions in binary ionic compounds. In these compounds, the metals generally take...
-
The standard enthalpy of formation of H2O(l) at 298 K is 285.8 kJ/ mol. Calculate the change in internal energy for the following process at 298 K and 1 atm: H2O(l) H2(g) + O2(g) Eo = ?
-
Consider the following galvanic cell: Calculate the Ksp value for Ag2SO4(s). Note that to obtain silver ions in the right compartment (the cathode compartment), excess solid Ag2SO4 was added and some...
-
See the option quote on IBM from the CBOE Web site on the next page showing options expiring in March and April 2022. a. Which option contract had the most trades that day? b. Which option contract...
-
Two European call options with a strike price of \($50\) are written on two different stocks. Suppose that tomorrow, the low-volatility stock will have a price of \($50\) for certain. The...
-
It is February 21, 2022, and you have decided to purchase 10 June call contracts on eBays stock with an exercise price of \($57.50.\) Because you are buying, you must pay the ask price. How much...

Study smarter with the SolutionInn App


