Read the corrosion potential of magnesium on the galvanic series in seawater below. The Pourbaix diagram...
Fantastic news! We've Found the answer you've been seeking!
Question:
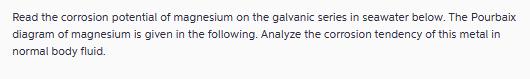

Transcribed Image Text:
Read the corrosion potential of magnesium on the galvanic series in seawater below. The Pourbaix diagram of magnesium is given in the following. Analyze the corrosion tendency of this metal in normal body fluid. Active Noble 0.5 Mild steel, cast iron Low alloy steel Aluminum alloys Cadmium 0.0 Zinc Berylium Platinum Graphite Austenitic Ni cast iron Aluminum bronze Naval brass, yellow brass, red brass Tin Copper Pd-Sn solder (50/50) Admiralty brass, aluminum brass Manganese bronze Silicon bronze Tin bronzes Stainless steel Types 410, 416 Nickel silver 90-10 Cu-Ni 80-20 Cu-Ni Stainless steel Type 430 Lead 70-30 Cu-Ni Ni-Al bronze Ni-Cr alloy 600 Silver braze alloys Nickel Silver | Stainless steel Type 304 Ni-Cu alloys 400, K-500 (Monel) Stainless steel Type 316 Alloy 20 | Ni-Fe-Cr alloy 825 Ni-Cr-Mo-Cu-Si alloy B Titanium | Ni-Cr-Mo alloy C Magnesium -0.5 -1.0 E in V vs. SCE -1.5 Read the corrosion potential of magnesium on the galvanic series in seawater below. The Pourbaix diagram of magnesium is given in the following. Analyze the corrosion tendency of this metal in normal body fluid. Active Noble 0.5 Mild steel, cast iron Low alloy steel Aluminum alloys Cadmium 0.0 Zinc Berylium Platinum Graphite Austenitic Ni cast iron Aluminum bronze Naval brass, yellow brass, red brass Tin Copper Pd-Sn solder (50/50) Admiralty brass, aluminum brass Manganese bronze Silicon bronze Tin bronzes Stainless steel Types 410, 416 Nickel silver 90-10 Cu-Ni 80-20 Cu-Ni Stainless steel Type 430 Lead 70-30 Cu-Ni Ni-Al bronze Ni-Cr alloy 600 Silver braze alloys Nickel Silver | Stainless steel Type 304 Ni-Cu alloys 400, K-500 (Monel) Stainless steel Type 316 Alloy 20 | Ni-Fe-Cr alloy 825 Ni-Cr-Mo-Cu-Si alloy B Titanium | Ni-Cr-Mo alloy C Magnesium -0.5 -1.0 E in V vs. SCE -1.5
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these general management questions
-
Consider the following table showing hypothetical balance-of-payments data for the United States. Complete the table by selecting the correct value for each missing entry Current Account U.S....
-
The corrosion potential of iron immersed in a de-aerated acidic solution of pH = 3 is -0.720 V as measured at 25C relative to the standard calomel electrode with potential 0.2802 V. A Tafel plot of...
-
Assume you are considering opening a retail business. You are trying to decide whether to have a traditional brick-and-mortar store or to sell only online. Explain how the activities and costs differ...
-
Why is limited liability more important for firms trying to raise funds from a large number of investors than for firms trying to raise funds from a small number of investors?
-
Stephens, Atkins, and Kingston (2009) conducted a research study demonstrating that swearing can help reduce pain. In the study, each participant was asked to plunge a hand into icy water and keep it...
-
What is the difference between object-oriented languages and UML?
-
A sale of merchandise on account for $12,200 is subject to a 6% sales tax. (a) Should the sales tax be recorded at the time of sale or when payment is received? (b) What is the amount of the sale?...
-
Problem 1. Consider the Fibonacci numbers, define recursively by F = 0, F = 1, and Fn = Fn1 + Fn-2 for all n 2; so the first few terms are 0, 1, 1, 2, 3, 5, 8, 13, .... Fn For all n >2, define the...
-
The information provided in this test allows you to set up the Cieneral, Payables and Receivables ledgers for an Ontario company named CHRIS'S WELDING owned and run by Chris George, amaster welder....
-
On October 3, Sandberg Art Supplies did an inventory and determined that they were running low on several Schutzen products that had been greatly in demand for the previous month. Their clerk then...
-
Consider diffusion in a spherical particle governed by J cs r with boundary conditions and c t = Ds Cs c r cs = r + 2 . 2 (R t) = 3/1/ r r (0, t) = 0, where D, is the diffusion coefficient, R is the...
-
Calculate the time (in seconds) required for the concentration of A to decrease from 1.0 M to 0.45 M. 1/[A] 120 100 80 60 40 20 0 0 20 K y = 0.7957x + 61.714 R = 0.9998 40 Time (sec) O 60
-
If you look up the radius of the Cl atom in various online sources, you will find values ranging from 79 to 182 pm (picometers; 1 pm = 1 10- m). The most common values are 100 pm and 182 pm. What can...
-
What are the correct units for k? 000 O L/(mol.h) mol/(L. h) (h. mol)/L (hL)/mol
-
In a colloidal dispersion solute particles are large enough to be discernible by the naked eye. (1) (2) (3) The selectivity of a semi-permeable membrane is based on polarity differences among...
-
Nancy Koehn's new case on the rebirth of Starbucks under Howard Schultz "distills 20 years of my thinking about the most important lessons of strategy, leadership, and managing in turbulence."...
-
What are the main distinctions between the different schools of legal interpretation?
-
The limiting molar conductivities of NaI, NaCH3CO" and Mg(CH3C02)2 are 12.69 mS m2 mol-1, 9.10 mS m2 mol-1, and 18.78 mS m2 mol-1, respectively (all at 25C). What is the limiting molar conductivity...
-
Calculate the maximum non-expansion work per mole that may be obtained from a fuel cell in which the chemical reaction is the combustion of propane at 298 K.
-
The group C4v consists of the elements E, 2C4, C2, and 2v, 2d construct the group multiplication table.
-
The following are summaries of the cash book and bank accounts of P. Maclaran who does not keep her books using the double entry system. You are required to: (a) Calculate the value of P. Maclaran's...
-
Jean Smith, who retails wooden ornaments, has been so busy since she commenced business on 1 April 2015 that she has neglected to keep adequate accounting records. Jean's opening capital consisted of...
-
The following trial balance of the Grampian Golf Club was extracted from the books as on 31 December 2016: (1) Bar purchases and sales were on a cash basis. Bar inventory at 31 December 2016 was...

Study smarter with the SolutionInn App


