Reagent: A 1) Na 2) Met 3) 9-BBN 4) HO, NaOH 1) 9-6BN 2) HO, NaOH...
Fantastic news! We've Found the answer you've been seeking!
Question:
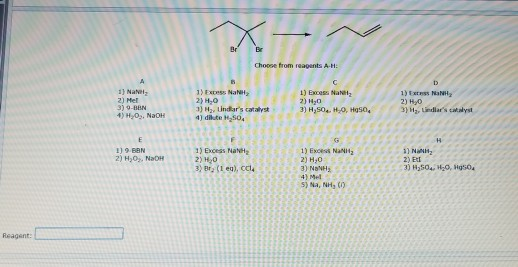
Transcribed Image Text:
Reagent: A 1) Na 2) Met 3) 9-BBN 4) H₂O₂, NaOH 1) 9-6BN 2) H₂O₂, NaOH X Br Br Choose from reagents A-H: B 1) Excess Na 2) H₂0 3) H₂, Lindlar's catalyst 4) dikute H₂50, 1) Exors NaN H 2) H₂0 3) Br, (1 eq), CCl4 C 1) Excess Na 2) H₂0 3) H₂50 H₂O, Hg50, 1) Exors NaN 2) H₂0 3) MẠNH, 4) Mel 5) Na, NH₂ (1) D 1) Extrus NINH 2) H₂0 3) H₂, Lindlar's catalyst H 1) Na 2) Et 3) H₂50₁, H₂O, Hg50₂ Reagent: A 1) Na 2) Met 3) 9-BBN 4) H₂O₂, NaOH 1) 9-6BN 2) H₂O₂, NaOH X Br Br Choose from reagents A-H: B 1) Excess Na 2) H₂0 3) H₂, Lindlar's catalyst 4) dikute H₂50, 1) Exors NaN H 2) H₂0 3) Br, (1 eq), CCl4 C 1) Excess Na 2) H₂0 3) H₂50 H₂O, Hg50, 1) Exors NaN 2) H₂0 3) MẠNH, 4) Mel 5) Na, NH₂ (1) D 1) Extrus NINH 2) H₂0 3) H₂, Lindlar's catalyst H 1) Na 2) Et 3) H₂50₁, H₂O, Hg50₂
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
x / 9 1 / 3 = 2 / 9
-
A steel ball of mass m = 50 g falls from the height h = 1.0 m on the horizontal surface of a massive slab. Find the cumulative momentum that the ball imparts to the slab after numerous bounces, if...
-
5 / 4 + 9 / 2x = 3 / 8 x 1 / 4
-
In the context of channel-of-distribution structures, discuss the distinguishing features of the Japanese distribution structure. Now, explain why the Japanese have not embraced the U.S distribution...
-
DBMSs are a component of every enterprise application, such as a supply chain management system. Why?
-
Two identical beads each have a mass m and charge q. When placed in a hemispherical bowl of radius R with frictionless, non-conducting walls, the beads move, and at equilibrium they are a distance R...
-
What do you know about the mathematical value of a project's internal rate of return under each of the following conditions? a. The present worth of the project is greater than 0 . b. The present...
-
Private College Transactions. Elizabeth College, a small private college, had the following transactions in fiscal year 2011. 1. Billings for tuition and fees totaled $5,600,000. Tuition waivers and...
-
Block 1 m m Block 2 Note: Figure not drawn to scale. Two blocks are connected by a string of negligible mass that passes over massless pulleys that turn with negligible friction, as shown in the...
-
isabelle sterling, the partner you work for, unexpectedly had to travel to Hong Kong. She left you the Tang Aluminum Products file for your client, Global Bank. Global will be lending Tang 1.9...
-
Consider an open economy characterized by the following equations: C = Co+c(Y-T) I = do +dY IM = m Y X = xY* Given m = 0.4, x = 0.3, and c + d = 0.6, what is the change in equilibrium output when...
-
What is a dissertation? What will it be like for you to write a dissertation? What excites you about the process? What challenges do you expect to encounter? Note: If you are not writing a...
-
15. Use the properties of continuity to determine the set of points at which each of the following functions is continuous. Justify your answers. a. The function f defined by f(x,y) b. The function g...
-
Here are the mercury concentration of mercury (in mg/kg) found in fish captured from the 53 lakes and rivers of Florida. The CDC has established that a concentration greater than 2ug/kg is considered...
-
Let V be a vector space of dimension n, and let W, W2 be subspaces of V. (a) Assume that W W = {0}. Show that dim(W W) = dim(W) + dim(W). ( where the direct sum W W of two subspaces W and W of a...
-
What are the stages of team development. . Define each stage and list two (2) characteristics of each stage. How can does the two stages work together for Human Resources when training managers.
-
You bought 250 shares of McWeber Inc. stock on margin. Price paid was $75 per share. The initial margin is 60%. The maintenance margin is 25%. Assume the price did not fall. Instead the price...
-
One hundred pounds of water at atmospheric pressure are heated from 60F to 200F. What is the enthalpy change? The internal energy change? Why is the difference between the internal energy change and...
-
Propose a mechanism for the following reaction. Show the structure of the compound that results from hydrolysis and decarboxylation of the product. CHO + CH2(COOCH2CH3)2 hydrolysis, decarboxylation...
-
An unknown reducing disaccharide is found to be unaffected by invertase enzymes. Treatment with an -galactosidase cleaves the disaccharide to give one molecule of D-fructose and one molecule of...
-
Without looking back, propose a mechanism for the hydrolysis of this iminium salt to the alkylated ketone. The first step is attack by water, followed by loss of a proton to give a carbinolamine....
-
Consider the dynamic system, a mass, spring and damper structure, shown in Figure 2.2. (a) Draw a free-body diagram for the dynamic system (b) Derive the Input-Output model of the dynamic system k...
-
Two connected cars with an applied input force \(u(t)\) and negligible rolling friction can be represented by a translational mechanical system as shown below. (a) Draw the free-body diagrams of this...
-
Consider an RLC circuit consisting of a resistor \((R)\), an inductor \((L)\), and a capacitor \((C)\), connected in series, as depicted in Figure 2.4. Derive the Input-Output model of the network....

Study smarter with the SolutionInn App



