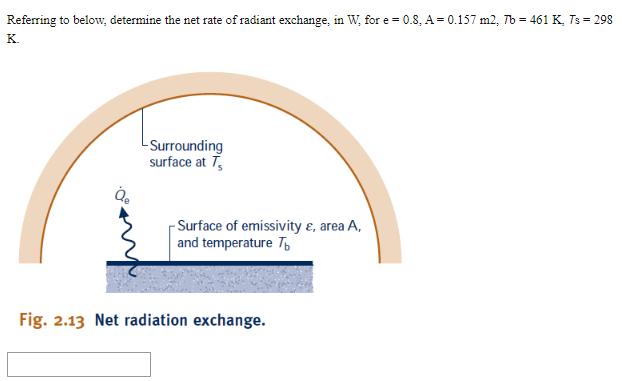
Referring to below, determine the net rate of radiant exchange, in W, for e=0.8, A =...
Fantastic news! We've Found the answer you've been seeking!
Question:



Transcribed Image Text:
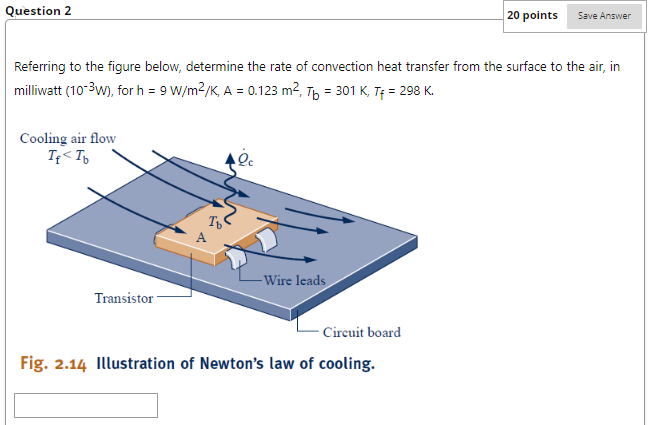
Referring to below, determine the net rate of radiant exchange, in W, for e=0.8, A = 0.157 m2, Tb = 461 K, Ts = 298 K. -Surrounding surface at T -Surface of emissivity &, area A, and temperature T Fig. 2.13 Net radiation exchange. Question 2 Cooling air flow If Question 3 20 points Save Answer A gas is compressed in a piston-cylinder assembly from p = 2.2 bar to p2 = 7.0 bar, V = 0.02 m in a process during which the relation between pressure and volume is pv1.4 = constant. The mass of the gas is 0.4 kg. If the specific internal energy of the gas increases by 40 kJ/kg during the process, determine the heat transfer, in Joules. Kinetic and potential energy changes are negligible. Question 4 20 points Save Answer A mass of 10 kg undergoes a process during which there is heat transfer from the mass at a rate of 5 kJ per kg, an elevation decrease of 47 m, and an increase in velocity from 15 m/s to 27 m/s. The specific internal energy decreases by 5 kJ/kg and the acceleration of gravity is constant at 9.7 m/s2. Determine the work for the process, in Joules. Question 5 20 points Evaluate the overall work, in kJ. Save Answer Carbon monoxide gas (CO) contained within a piston- cylinder assembly undergoes three processes in series: Process 1-2: Expansion from p = 3.7 bar, V = 0.9 m to V = 1.2 m, during which the pressure-volume relationship is pV = constant. Process 2-3: Constant-volume heating from state 2 to state 3, where p3 = 3.7 bar. Process 3-1: Constant-pressure compression to the initial state. Referring to below, determine the net rate of radiant exchange, in W, for e=0.8, A = 0.157 m2, Tb = 461 K, Ts = 298 K. -Surrounding surface at T -Surface of emissivity &, area A, and temperature T Fig. 2.13 Net radiation exchange. Question 2 Cooling air flow If Question 3 20 points Save Answer A gas is compressed in a piston-cylinder assembly from p = 2.2 bar to p2 = 7.0 bar, V = 0.02 m in a process during which the relation between pressure and volume is pv1.4 = constant. The mass of the gas is 0.4 kg. If the specific internal energy of the gas increases by 40 kJ/kg during the process, determine the heat transfer, in Joules. Kinetic and potential energy changes are negligible. Question 4 20 points Save Answer A mass of 10 kg undergoes a process during which there is heat transfer from the mass at a rate of 5 kJ per kg, an elevation decrease of 47 m, and an increase in velocity from 15 m/s to 27 m/s. The specific internal energy decreases by 5 kJ/kg and the acceleration of gravity is constant at 9.7 m/s2. Determine the work for the process, in Joules. Question 5 20 points Evaluate the overall work, in kJ. Save Answer Carbon monoxide gas (CO) contained within a piston- cylinder assembly undergoes three processes in series: Process 1-2: Expansion from p = 3.7 bar, V = 0.9 m to V = 1.2 m, during which the pressure-volume relationship is pV = constant. Process 2-3: Constant-volume heating from state 2 to state 3, where p3 = 3.7 bar. Process 3-1: Constant-pressure compression to the initial state.
Expert Answer:

Related Book For 

Principles of heat transfer
ISBN: 978-0495667704
7th Edition
Authors: Frank Kreith, Raj M. Manglik, Mark S. Bohn
Posted Date:
Students also viewed these mechanical engineering questions
-
A 0.61 m radius hemisphere (811 K surface temperature) is filled with a gas mixture at 533 K and 2-atm pressure containing 6.67 percent CO2 and water vapor at 0.5 percent relative humidity. Determine...
-
During 2018, Susan incurred and paid the following expenses for Beth (her daughter), Ed (her father), and herself: Surgery for...
-
Paulsen Corporation makes two products, W and P, in a joint process. At the split-off point, 50,000 units of W and 60,000 units of P are available each month. Monthly joint production costs are...
-
A dealer's profit, in units of $5000, on a new automobile is given by Y = X2, where X is a random variable having the density function(a.) Find the probability density function of the random variable...
-
What limits the lifetime of a battery?
-
Part One: In 2012, Penny Henderson opened Pennys Posies, a small retail shop selling floral arrangements. On December 31, 2013, her accounting records show the following: Sales...
-
(a) Equity of KGF Ltd. (KGFL) is Rs. 410 Crores, its debt, is worth Rs. 170 Crores. Printer Division segments value is attributable to 74%, which has an Asset Beta (p) of 1.45, balance value is...
-
Corrie wants to earn a net operating income of $10,000 this month. She currently sells her product for $50 each, has $10 of variable expense per product, and incurs $5,000 of fixed expenses. How many...
-
Public entities are required to report on a quarterly basis to their Executive Authority. Treasury Regulations 5.3.1, 29.3.1 and 30.2.1 require the accounting officer of an institution and accounting...
-
P(B|E) Find the indicated probabilities by referring to the given tree diagram and by using Bayess Rule. A P(A) = 0.2 P(E|A)=0.6 E P(ECA) = 0.4 -EC P(B) = 0.55 P(E|B)=0.7 -E B EC P(E|B) = 0.3 P(C) =...
-
The weather forecast says there is a 40% chance of rain today and a 30% chance of rain tomorrow. Assume the days are independent. (a) What is the chance of rain on both days? (b) What is the chance...
-
P(B|E c ) Find the indicated probabilities by referring to the given tree diagram and by using Bayess Rule. A P(A) = 0.2 P(E|A)=0.6 E P(ECA) = 0.4 -EC P(B) = 0.55 P(E|B)=0.7 -E B EC P(E|B) = 0.3 P(C)...
-
Use the information given in Problem 22 to find: (a) P(A 1 |E) (b) P(A 2 |E) Data from Problem 22 Suppose that events A 1 and A 2 form a partition of the sample space S with P(A 1 ) = 0.35 and P(A 2...
-
Historically, victims of the Ebola virus had a 30% chance of survival. (a) A new treatment is tested on three victims, and at least two of them survive. What is the probability of this happening, if...
-
The weight of the delta function in the power spectral density (PSD) (the dc value) of a random process X(t) that has the correlation function given by RX(t) = 4[1 + exp(-2A|T|)] is O a. 3 m /2 O b....
-
(a) Explain why the concentration of dissolved oxygen in freshwater is an important indicator of the quality of the water. (b) How is the solubility of oxygen in water affected by increasing...
-
A Trombe wall is a masonry wall often used in passive solar homes to store solar energy. Suppose such a wall, fabricated from 20 cm thick solid concrete blocks (k = 0.13 W/(mK), = 0.05 \ 10 5 m 2 /s...
-
Wearing layers of clothing in cold weather is often recommended because dead-air spaces between the layers keep the body warm. The explanation for this is that the heat loss from the body is less....
-
By substituting Equation 9.1 for Eb (T) in Equation 9.4 and performing the integration over the entire spectrum, derive a relationship between s and the constants C1 and C2 in Equation...
-
You are working for Microsoft evaluating the possibility of selling energy drinks. Microsofts WACC is 8.1%. Energy drinks would be a new line of business for Microsoft, however, so the systematic...
-
By using the yield to maturity on AT&Ts debt, we found that its pretax cost of debt is 3.65%. If AT&Ts tax rate is 25%, what is its effective cost of debt?
-
You are analyzing AT&Ts potential acquisition of Netflix. AT&T plans to offer \($150\) billion as the purchase price for Netflix, and it will need to issue additional debt and equity to finance such...

Study smarter with the SolutionInn App


