A reaction profile (not to scale!) for the reaction 03 + 0 2 0 is shown...
Fantastic news! We've Found the answer you've been seeking!
Question:
![A reaction profile (not to scale!) for the reaction [ mathrm{O}_{3}+mathrm{O} longrightarrow 2 mathrm{O}_{2} ] is shown](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2022/09/63198c52ea3c2_1662618703658.jpg)
![[ mathrm{O}_{3}+mathrm{O} rightarrow 2 mathrm{O}_{2} ] is shown below: [ E(k J) ] Which of the following are true? (S](https://dsd5zvtm8ll6.cloudfront.net/si.experts.images/questions/2022/09/63198c530efb2_1662618703844.jpg)
Transcribed Image Text:
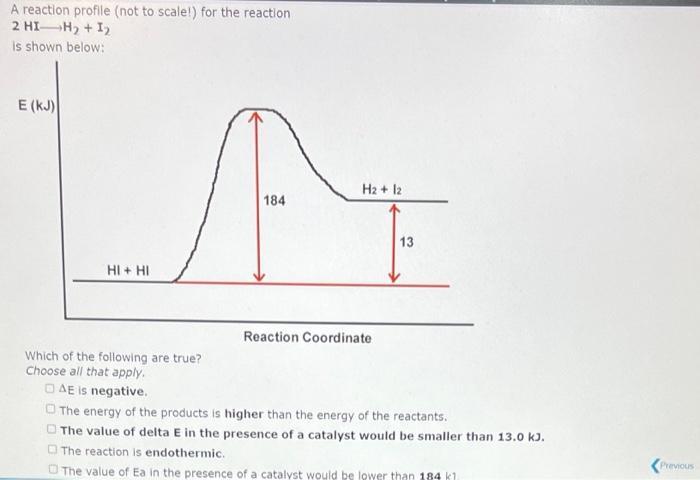
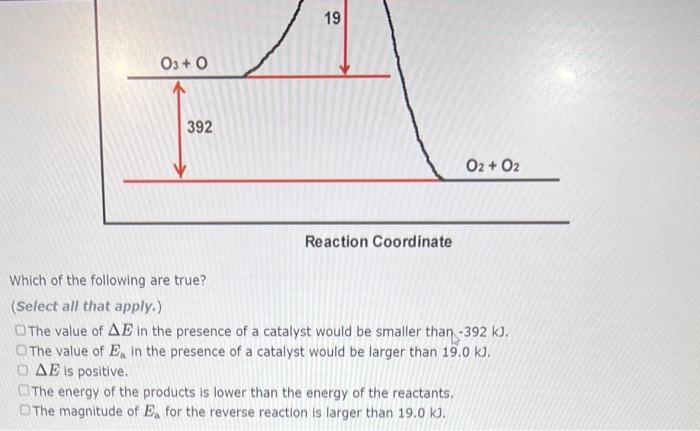
A reaction profile (not to scale!) for the reaction 03 + 0 2 0₂ is shown below: E (KJ) 03+0 392 19 Reaction Coordinate This reaction is (exothermic,endothermic) The value of the activation energy for this reaction is 02 + 02 kJ and the value of AE is kJ. A reaction profile (not to scale!) for the reaction 2 HI-H₂ + 1₂ is shown below: E (KJ) HI+ HI Which of the following are true? Choose all that apply. AE is negative. 184 H2 + 12 Reaction Coordinate 13 The energy of the products is higher than the energy of the reactants. The value of delta E in the presence of a catalyst would be smaller than 13.0 kJ. The reaction is endothermic. The value of Ea in the presence of a catalyst would be lower than 184 kl Previous 03+0 20₂ is shown below: E (KJ) 03+0 392 19 Reaction Coordinate 02 + 02 Which of the following are true? (Select all that apply.) The value of AE in the presence of a catalyst would be smaller than -392 kJ. The value of E, in the presence of a catalyst would be larger than 19.0 kJ. DAE is positive. The energy Show Hint ducts is lower than the energy of the reactants. Pie 03+0 392 19 Reaction Coordinate 02 + 02 Which of the following are true? (Select all that apply.) The value of AE in the presence of a catalyst would be smaller than -392 kJ. The value of E, in the presence of a catalyst would be larger than 19.0 kJ. OAE is positive. The energy of the products is lower than the energy of the reactants. The magnitude of E, for the reverse reaction is larger than 19.0 kJ. A reaction profile (not to scale!) for the reaction 03 + 0 2 0₂ is shown below: E (KJ) 03+0 392 19 Reaction Coordinate This reaction is (exothermic,endothermic) The value of the activation energy for this reaction is 02 + 02 kJ and the value of AE is kJ. A reaction profile (not to scale!) for the reaction 2 HI-H₂ + 1₂ is shown below: E (KJ) HI+ HI Which of the following are true? Choose all that apply. AE is negative. 184 H2 + 12 Reaction Coordinate 13 The energy of the products is higher than the energy of the reactants. The value of delta E in the presence of a catalyst would be smaller than 13.0 kJ. The reaction is endothermic. The value of Ea in the presence of a catalyst would be lower than 184 kl Previous 03+0 20₂ is shown below: E (KJ) 03+0 392 19 Reaction Coordinate 02 + 02 Which of the following are true? (Select all that apply.) The value of AE in the presence of a catalyst would be smaller than -392 kJ. The value of E, in the presence of a catalyst would be larger than 19.0 kJ. DAE is positive. The energy Show Hint ducts is lower than the energy of the reactants. Pie 03+0 392 19 Reaction Coordinate 02 + 02 Which of the following are true? (Select all that apply.) The value of AE in the presence of a catalyst would be smaller than -392 kJ. The value of E, in the presence of a catalyst would be larger than 19.0 kJ. OAE is positive. The energy of the products is lower than the energy of the reactants. The magnitude of E, for the reverse reaction is larger than 19.0 kJ.
Expert Answer:
Answer rating: 100% (QA)
Q1 Q2 Q3 In the given energy profile 19 kj represents the activation energy Ea ... View the full answer

Posted Date:
Students also viewed these computer network questions
-
Find dy/dx and d2 y/dx2. x = t + sin t, y = t - cos t
-
Find a linear function h given h(1) = 4 and h( -2) = 13. Then find h(2). (a) (0, -3) (b) (- 1/4, 7) (c) (2/11, - 1) (d) (0.03, 0)
-
Find a linear function h given h(1) = 4 and h(-2) = 13. Then find h(2).
-
The balance sheet of Lamont Bros. follow: (a) What portions of Lamont's assets were provided by debt, contributed capital, and earned capital? Reduce contributed capital by the cost of the treasury...
-
A 75.0-kg wrecking ball hangs from a uniform heavy-duty chain having a mass of 26.0 kg. (a) Find the maximum and minimum tension in the chain. (b) What is the tension at a point three fourths of the...
-
You must get from a point P on the straight shore of a lake to a stranded swimmer who is 50 m from a point Q on the shore that is 50 m from you (see figure). If you can swim at a speed of 2 m/s and...
-
Interactive video games and physical fitness. Wii Fit is an interactive video game marketed to consumers who want to increase their physical fitness level. The effectiveness of Wii Fit activities...
-
The bookkeeper of Beautiful World Landscaping prepared the company's balance sheet while the accountant was ill. The balance sheet contains numerous errors. In particular, the bookkeeper knew that...
-
Wexpro, incorporated produces several products from processing 1 ton of clypton, a rare mineral. Material and processin $63,000 per ton, one-fourth of which is allocated to product X15. Six thousand...
-
these are the answer sheet please fill this XY Consumer Products GENERAL JOURNAL GJ01 Date General Ledger 2003 Description Acc # Debit Credit Mar. 31 Bank Charges & Interest Mar. 31 Employers Payroll...
-
Use nodal analysis to find V in the circuit of figure shown below. 202 402 www 120 15 V 6 Z30 A 30 2 soa
-
The Matsui Lubricants plant uses the weighted-average method to account for its work-in-process inventories. The accounting records show the following information for a particular day: Beginning WIP...
-
James Cook, a production department worker, is paid on hourly basis at a rate of $15 per hour. James works 40 hours per week. Any time James works over 40 hours, it is considered as overtime and he...
-
You just started working as a Health Service Manager within one of the following healthcare industries. First, choose an industry below to discuss the questions that follow: Ambulatory Surgery center...
-
Q10: Region ( experienced compressive stresses and has a than the rest of the bracket. Region ( ) experienced tension stresses and has a of the bracket. Deep Drawing and Stretch Forming width (into...
-
A sample of 1500 computer chips revealed that 32% of the chips do not fail in the first 1000 hours of their use. The company\'s promotional literature claimed that above 29% do not fail in the first...
-
Spice Company has two divisions, Parsley and Sage. Parsley produces a unit that Sage could use in its production Sage currently is purchasing 51.000 units from an outside supplier for $60.00. Parsley...
-
Fill in each blank so that the resulting statement is true. 83 + 103 = ______ .
-
Describe the difference between accounting for governmental funds and proprietary funds. AppendixLO1
-
What is the difference between an enterprise fund and an internal service fund? AppendixLO1
-
What is the difference between a permanent fund and a private-purpose trust fund? AppendixLO1

Study smarter with the SolutionInn App

