Question: The following selected account balances were taken from ABC Company's general ledgers for July: Inventory Notes payable Land Retained earnings Accounts receivable Salaries payable
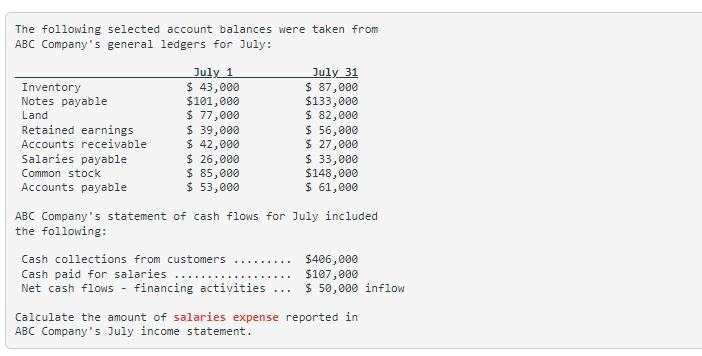
The following selected account balances were taken from ABC Company's general ledgers for July: Inventory Notes payable Land Retained earnings Accounts receivable Salaries payable Common stock Accounts payable July 1 $ 43,000 $101,000 $ 77,000 $ 39,000 $ 42,000 $ 26,000 $ 85,000 $ 53,000 ABC Company's statement of cash flows for July included the following: Cash collections from customers Cash paid for salaries .... Net cash flows financing activities ... July 31 $ 87,000 $133,000 $ 82,000 $ 56,000 $ 27,000 $ 33,000 $148,000 $ 61,000 .... $406,000 $107,000 $50,000 inflow Calculate the amount of salaries expense reported in ABC Company's July income statement.
Step by Step Solution
There are 3 Steps involved in it
First lets determine the change in salaries payable from the beginning of the month to the end of th... View full answer

Get step-by-step solutions from verified subject matter experts


