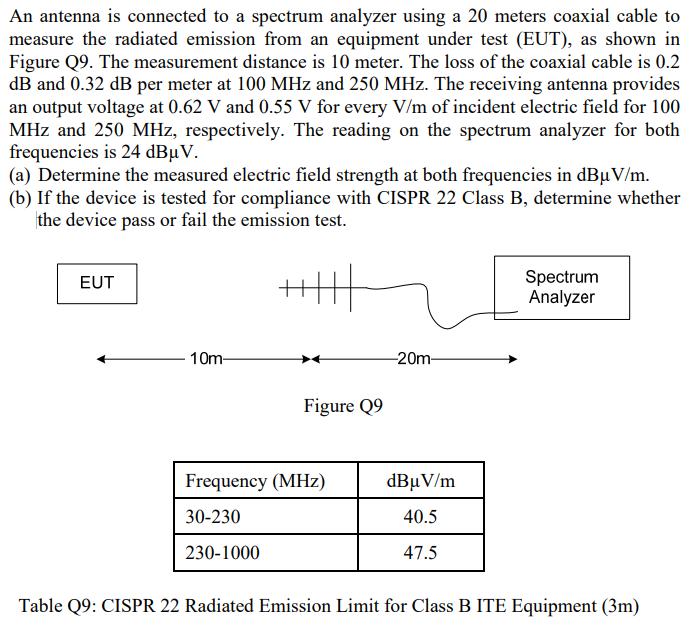
An antenna is connected to a spectrum analyzer using a 20 meters coaxial cable to measure...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
An antenna is connected to a spectrum analyzer using a 20 meters coaxial cable to measure the radiated emission from an equipment under test (EUT), as shown in Figure Q9. The measurement distance is 10 meter. The loss of the coaxial cable is 0.2 dB and 0.32 dB per meter at 100 MHz and 250 MHz. The receiving antenna provides an output voltage at 0.62 V and 0.55 V for every V/m of incident electric field for 100 MHz and 250 MHz, respectively. The reading on the spectrum analyzer for both frequencies is 24 dBμV. (a) Determine the measured electric field strength at both frequencies in dBµV/m. (b) If the device is tested for compliance with CISPR 22 Class B, determine whether the device pass or fail the emission test. EUT 10m- Figure Q9 Frequency (MHz) 30-230 230-1000 -20m- dBμV/m 40.5 47.5 Spectrum Analyzer Table Q9: CISPR 22 Radiated Emission Limit for Class B ITE Equipment (3m) An antenna is connected to a spectrum analyzer using a 20 meters coaxial cable to measure the radiated emission from an equipment under test (EUT), as shown in Figure Q9. The measurement distance is 10 meter. The loss of the coaxial cable is 0.2 dB and 0.32 dB per meter at 100 MHz and 250 MHz. The receiving antenna provides an output voltage at 0.62 V and 0.55 V for every V/m of incident electric field for 100 MHz and 250 MHz, respectively. The reading on the spectrum analyzer for both frequencies is 24 dBμV. (a) Determine the measured electric field strength at both frequencies in dBµV/m. (b) If the device is tested for compliance with CISPR 22 Class B, determine whether the device pass or fail the emission test. EUT 10m- Figure Q9 Frequency (MHz) 30-230 230-1000 -20m- dBμV/m 40.5 47.5 Spectrum Analyzer Table Q9: CISPR 22 Radiated Emission Limit for Class B ITE Equipment (3m) An antenna is connected to a spectrum analyzer using a 20 meters coaxial cable to measure the radiated emission from an equipment under test (EUT), as shown in Figure Q9. The measurement distance is 10 meter. The loss of the coaxial cable is 0.2 dB and 0.32 dB per meter at 100 MHz and 250 MHz. The receiving antenna provides an output voltage at 0.62 V and 0.55 V for every V/m of incident electric field for 100 MHz and 250 MHz, respectively. The reading on the spectrum analyzer for both frequencies is 24 dBμV. (a) Determine the measured electric field strength at both frequencies in dBµV/m. (b) If the device is tested for compliance with CISPR 22 Class B, determine whether the device pass or fail the emission test. EUT 10m- Figure Q9 Frequency (MHz) 30-230 230-1000 -20m- dBμV/m 40.5 47.5 Spectrum Analyzer Table Q9: CISPR 22 Radiated Emission Limit for Class B ITE Equipment (3m) An antenna is connected to a spectrum analyzer using a 20 meters coaxial cable to measure the radiated emission from an equipment under test (EUT), as shown in Figure Q9. The measurement distance is 10 meter. The loss of the coaxial cable is 0.2 dB and 0.32 dB per meter at 100 MHz and 250 MHz. The receiving antenna provides an output voltage at 0.62 V and 0.55 V for every V/m of incident electric field for 100 MHz and 250 MHz, respectively. The reading on the spectrum analyzer for both frequencies is 24 dBμV. (a) Determine the measured electric field strength at both frequencies in dBµV/m. (b) If the device is tested for compliance with CISPR 22 Class B, determine whether the device pass or fail the emission test. EUT 10m- Figure Q9 Frequency (MHz) 30-230 230-1000 -20m- dBμV/m 40.5 47.5 Spectrum Analyzer Table Q9: CISPR 22 Radiated Emission Limit for Class B ITE Equipment (3m)
Expert Answer:
Answer rating: 100% (QA)
To determine the measured electric field strength at both frequencies we need to first account for the cable loss and then convert the voltage measure... View the full answer

Related Book For 

Posted Date:
Students also viewed these mathematics questions
-
A garden hose is held as shown in Figure P9.13. The hose is originally full of motionless water. What additional force is necessary to hold the nozzle stationary after the water flow is turned on, if...
-
As shown in Figure P18.42, water is pumped into a tall vertical cylinder at a volume flow rate R. The radius of the cylinder is r, and at the open top of the cylinder a tuning fork is vibrating with...
-
A spring exhibits a force-versus-displacement characteristic as shown in Figure E2.11. For small deviations from the operating point x0, find the spring constant when x0 is (a) -1.4; (b) 0; (c) 3.5....
-
9) The mole (Avogadro number N) is defined as the number of atoms in exactly 12 grams of 2C, calculate the: a) number of atoms in 2 g of C b) number of atoms in 5 g of C
-
Using the same information as In Problem, develop the best policies for a periodic review system. A company begins a review of ordering policies [or its continuous review system by checking the...
-
Data mining is the process of using sophisticated statistical techniques to extract and analyze data from large databases to discern patterns and trends that were not previously known. The patterns...
-
Superhero Industries reported the following at September 30: Requirements 1. Record retirement of half of the bonds on October 1 at the call price of 101. 2. Record conversion of the remainder of the...
-
Refer to the financial statements for Castile Products, Inc., in Exercise 16-7. Assets at the beginning of the year totaled $280,000, and the stockholders equity totaled $161,600. Required: Compute...
-
write down three complete business rules describing relationships from the below entity relationship diagram. Each rule should fully describe the multiplicities of the relationship
-
The COVID-19 pandemic affected companies in the food industry in unique ways, particularly during 2020. Ruth's Hospitality Group (Ticker: RUTH) develops and operates fine dining restaurants under the...
-
Write an essay that discusses why oral communication skills, listening and interpersonal abilities skills are considered so important within an organization that wants to enhance its service...
-
Learning the language of business Match each of the numbered descriptions 1 through 7 with the term or phrase it best reflects. Indicate your answer by writing the letter for the term or phrase in...
-
Why does the sample size of a substantive test increase as the expected misstatement in the population increases?
-
State the objectives in the audit of cash.
-
Turner Valley Hospital plans to install a database management system, Hosp Info, that will maintain patient histories, including tests performed and their results, vital statistics, and medical...
-
In classical statistical sampling, what four items are used to obtain the optimum sample size for estimating the dollar amount of a population?
-
For each of the following independent cases (A to E), compute the missing values in the table: Case A B C D E Prime Cost Conversion Cost $ 9,450 $ 16,360 $ 56,900 110,300 48,650 Direct Materials...
-
Why do CPA firms sometimes use a combination of positive and negative confirmations on the same audit?
-
Apply the following operations on the corresponding polynomials: a. (x 3 + x 2 + x + 1) + (x 4 + x 2 + x + 1) b. (x 3 + x 2 + x + 1) (x 4 + x 2 + x + 1) c. (x 3 + x 2 ) (x 4 + x 2 + x + 1) d. (x 3...
-
An AP in a wireless network plays the same role as a link-layer switch in a wired network. However, a link-layer switch has no MAC address, but an AP normally needs a MAC address. Explain the reason.
-
Answer the following questions about a T-1 line: a. What is the duration of a frame? b. What is the overhead (number of extra bits per second)?
-
Justify the statement: Osmosis is of paramount importance in biological systems.
-
The freezing point of pure benzene is \(5.44^{\circ} \mathrm{C}\) and that of the solution containing \(2.092 \mathrm{~g}\) of benzaldehyde in \(100 \mathrm{~g}\) of benzene is \(4.44^{\circ}...
-
The molality of dissolved gases in water at \(0^{\circ} \mathrm{C}\) and \(1 \mathrm{~atm}\) is \(1.29 \times 10^{-3}\). The decrease in volume during melting of ice is \(0.0907 \mathrm{cc} /...

Study smarter with the SolutionInn App


