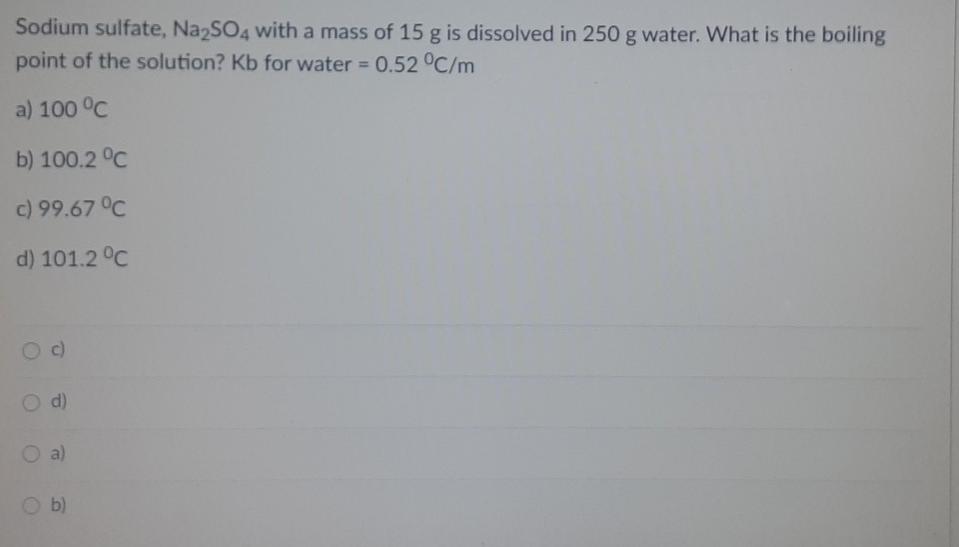
Sodium sulfate, Na2SO4 with a mass of 15 g is dissolved in 250 g water. What...
Fantastic news! We've Found the answer you've been seeking!
Question:


Transcribed Image Text:
Sodium sulfate, Na2SO4 with a mass of 15 g is dissolved in 250 g water. What is the boiling point of the solution? Kb for water = 0.52 °C/m a) 100 °C b) 100.2 °C c) 99.67 °C d) 101.2 °C c) d) O a) b) Fifty grams of an unknown nonelectrolyte solute is mixed with 2000 g of naphthalene (C10H8). The boiling point of the solution was observed to be equal to 80°C. If the boiling point elevation constant of naphthalene is 6.9 °C/m and the boiling point of pure naphthalene is 74.6°C: What is the boiling point elevation of the solution? a) 4 °C b) 5.0 °C c) 5.40 °C d) 5.50 °C a) b) Od) Fifty grams of an unknown nonelectrolyte solute is mixed with 2000 g of naphthalene (C10H8). The boiling point of the solution was observed to be equal to 80°C. If the boiling point elevation constant of naphthalene is 6.9 °C/m and the boiling point of pure naphthalene is 74.6°C: Compute for the molality of the solution. 0.0783 m O 0.783 m 0.0078 m 0.0782 m Sodium sulfate, Na2SO4 with a mass of 15 g is dissolved in 250 g water. What is the boiling point of the solution? Kb for water = 0.52 °C/m a) 100 °C b) 100.2 °C c) 99.67 °C d) 101.2 °C c) d) O a) b) Fifty grams of an unknown nonelectrolyte solute is mixed with 2000 g of naphthalene (C10H8). The boiling point of the solution was observed to be equal to 80°C. If the boiling point elevation constant of naphthalene is 6.9 °C/m and the boiling point of pure naphthalene is 74.6°C: What is the boiling point elevation of the solution? a) 4 °C b) 5.0 °C c) 5.40 °C d) 5.50 °C a) b) Od) Fifty grams of an unknown nonelectrolyte solute is mixed with 2000 g of naphthalene (C10H8). The boiling point of the solution was observed to be equal to 80°C. If the boiling point elevation constant of naphthalene is 6.9 °C/m and the boiling point of pure naphthalene is 74.6°C: Compute for the molality of the solution. 0.0783 m O 0.783 m 0.0078 m 0.0782 m
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
What is the boiling point of a solution of 0.150 g of glycerol, C3H8O3, in 20.0 g of water? What is the freezing point?
-
What is the boiling point of a solution made by adding 6.69 g of magnesium chloride to 243.3 g of water? Use the formula of the salt to obtain i.
-
A railroad car having a mass of 15 Mg is coasting at 1.5 m/s on a horizontal track. At the same time another car having a mass of 12 Mg is coasting at 0.75 m/s in the opposite direction. It the cars...
-
Consider the following graph. 2 7 6 C 10 10 H 11 E 12 15 14 (a). Find a minimum spanning tree of the graph using Kruskal's algorithm. List the edges in the order they are put into the tree. (b)....
-
Using the following financial information for Wilson National Bank, calculate as many of the liquidity indicators discussed in this chapter for Wilson as you can. Do you detect any significant...
-
A simplified version of the life cycle of grocery sacks is shown below. 4 In the late 1970s, supermarkets began to replace paper sacks with polyethylene (plastic) sacks. In the 1980s, a movement to...
-
Renkas Heaters selected data for October 2017 are presented here (in millions): Calculate the following costs: 1. Direct materials inventory 10/31/2017 2. Fixed manufacturing overhead costs for...
-
1. Complete Problem 17-36 using the FIFO method of process costing. In Problem 17-36, McKnight Handcraft is a manufacturer of picture frames for large retailers. Every picture frame passes through...
-
Westeros, Inc. receives scabbards (sheath for a sword) from two different suppliers. 70 percent of its scabbards come from Knights Watch, LLC, while 30 percent come from Crow, Inc. Records indicate...
-
There is an array A made of N integers. Your task is to choose as many integers from A as possible so that, when they are put in ascending order, all of the differences between all pairs of...
-
PROBLEM 1. Compute stress, strain and displacement at point A (x,y). Given: Elastic domain, E= 10 Mpa, v= 0.3 x= 4, y= 1.42 Fx= 47, Fy= 182.5 Each element is a 5x5 constant strain triangle, the...
-
On a patient in assist control what setting controls how easily the machine can be cycled?
-
If a bacterial culture experiences a tripling in size every four-hour interval, and the current estimated population stands at three million, what projection can be made regarding the population's...
-
Infocon, Inc. is planning to buy a suite of computers for its data center. The equipment will cost $22,500 and is expected to maintain the equipment at a cost of $6,250 per year. The equipment has a...
-
Consider a 2-year forward contract to buy 1 million shares of a non-dividend paying stock. The current stock price is $80 per share, the delivery price is $85 per share, and the continuously...
-
Micheal has written a report summarizing departmental projects and expenditures that will be read by executives and senior managers. He wants to the report to engage and inform. What type of format...
-
A quality characteristic is monitored by a control chart designed so the probability that a certain out-of-control condition will be detected on the first sample following the shift to that state is...
-
When an electric field is applied to a shallow bath of vegetable oil, why do tiny bits of thread floating in the oil align with the field like compasses in a magnetic field?
-
What is wrong with the following "proof" that every matrix with at least two rows is row equivalent to a matrix with a zero row? Perform R2 + R1 and R1 + R2. Now rows 1 and 2 are identical. Now...
-
Suppose that A is a 6 6 matrix with characteristic polynomial cA() = (1 + ) (1 - )2 (2 - )3. a. Prove that it is not possible to find three linearly independent vectors v1, v2, v3 in R6 such that...
-
In Exercises 1-3, a matrix A is given along with an iterate x5, produced as in Example 4.30. (a) Use these data to approximate a dominant eigenvector whose first component is 1 and a corresponding...
-
(a) The longitudinal data set "v4c" is in the vertical format; transform it into the horizontal format. (b) Transform the data set you obtained in part (a) back into the vertical format.
-
Perform some exploratory analysis on the DTS study described above. (a) Compute the mean and standard deviation of the HamD scores for the two treatment groups at each time point. (b) Treat repeated...
-
Plot the mean/SD of HIV knowledge of adolescent girls at baseline and three months post treatment stratified by treatment for the Sexual Health study.

Study smarter with the SolutionInn App



