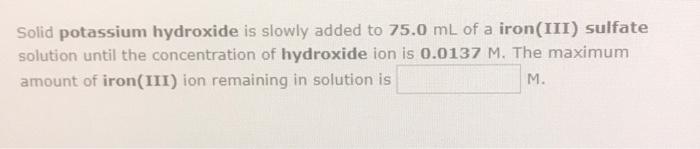
Solid potassium hydroxide is slowly added to 75.0 mL of a iron(III) sulfate solution until the...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Solid potassium hydroxide is slowly added to 75.0 mL of a iron(III) sulfate solution until the concentration of hydroxide ion is 0.0137 M. The maximum amount of iron(III) ion remaining in solution is M. Solid potassium hydroxide is slowly added to 75.0 mL of a iron(III) sulfate solution until the concentration of hydroxide ion is 0.0137 M. The maximum amount of iron(III) ion remaining in solution is M.
Expert Answer:
Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these chemistry questions
-
A 1.0 M Na2SO4 solution is slowly added to 10.0 mL of a solution that is 0.20 M in Ca2+ and 0.30 M in Ag+. (a) Which compound will precipitate first: CaSO4 (Ksp = 2.4 x 10-5) or Ag2SO4(Ksp = 1.5 x...
-
Solid NaI is slowly added to a solution that is 0.010 M in Cu1 and 0.010 M in Ag1. (a) Which compound will begin to precipitate first? (b) Calculate [Ag1] when CuI just begins to precipitate. (c)...
-
Solid zinc acetate is slowly added to 175 mL of a 0.0604 M sodium cyanide solution. The concentration of zinc ion required to just initiate precipitation is M. Submit Answer Try Another Version 9...
-
Which of the following questions involve microeconomics, and which involve macroeconomics? In each case, explain your answer. a. Why did consumers switch to smaller cars in 2008? b. Why did overall...
-
What is the price of a three-year, default-free security with a face value of $1000 and an annual coupon rate of 4%? What is the yield to maturity for thisbond? Maturity (years) Zero-coupon YTM 4.00%...
-
C&C Properties is an S corporation that owns two rental real estate undertakings: Carrot Plaza and Cantaloupe Place. Both properties produce an annual $10,000 operating loss. C&Cs Schedule K...
-
On January 1, Lowe, Inc., issued \$500,000 of ten percent, 20-year bonds for \(\$ 598,964\), yielding an effective interest rate of eight percent. Semiannual interest is payable on June 30 and...
-
Presented below is selected information for three regional divisions of Glendo Company. Instruction (a) Compute the return on investment for each division. (b) Compute the residual income for each...
-
Question 1 An investor purchases a call for CL0 = $4.10 with a strike price of XL = $49 and sells a call for CH0 = $0.90 with a strike price of XH = $55. 1) Identify and briefly explain option...
-
On October 1, 2020, Mary Graham organized a computer service company called Echo Systems. Echo is organized as a sole proprietorship and will provide consulting services, computer system...
-
Bond Refunding The Treasurer of Moribund Corp. has finished her bond refunding analysis and she just needs to calculate the final value. Using the following cash flows, determine the final Net...
-
5. LGI projects the following after-tax cash flows from operations from its aging Bowie, Maryland plant (which first went on line in 1953) over the next five years. What is the PV of these cash...
-
The dynamic system shown below consists of a cart of mass M carrying a simple pendulum L. The cart moves vertically. It is constrained by a spring k and damper c, and is subject to an external force...
-
You have the following information on Ralph's Company (1) The firm's bonds have a maturity of 20 years, an 8.00% annual coupon, a par value of $1,000, and a market price of $1,075.00.(2) The...
-
"Imagine you are an auditor tasked with reviewing the financial statements of a company. During your audit, you identify a significant matter that requires emphasis to ensure stakeholders are...
-
A company is attempting to immunize a schedule of liability cash flows via cash flow matching. The cash flows are as follows: Year Liability (000s) 1 126 2 272 3 372 Starting from the end and working...
-
When there is uneven distributions of grouped data within bins the effects will tend to out unless there is systematic skewness.
-
Fred Farmer needs to prepare a balance sheet for his bank. He spent the day getting the following information. Fred needs your help to build a balance sheet and evaluate it. The information was...
-
Determine the oxidation number for the indicated element in each of the following compounds: (a) Co in LiCoO2 (b) Al in NaAlH4 (c) C in CH3OH (methanol) (d) N in GaN (e) Cl in HClO2 (f) Cr in BaCrO4.
-
What is the name given to the kind of behavior demonstrated by a metal hydroxide in this graph? pH Solubility
-
If a substance is an Arrhenius base, is it necessarily a Brnsted- Lowry base? Is it necessarily a Lewis base? Explain.
-
Skyway Inc. identified a cost pool related to material handling costs for its manufacturing process. Using historical data and ordinary least squares regression, Skyway summarized two different...
-
Assume that a manufacturing company is determining the optimal minimum level of cash to maintain in a financial institution to cover short-term needs. Separately consider each of the following...
-
Prior to the Tax Cuts and Jobs Act of 2018 (TCJA), multinational firms domiciled in the United States did not have to pay U.S. income taxes on foreign earnings, unless those earnings were returned...

Study smarter with the SolutionInn App



