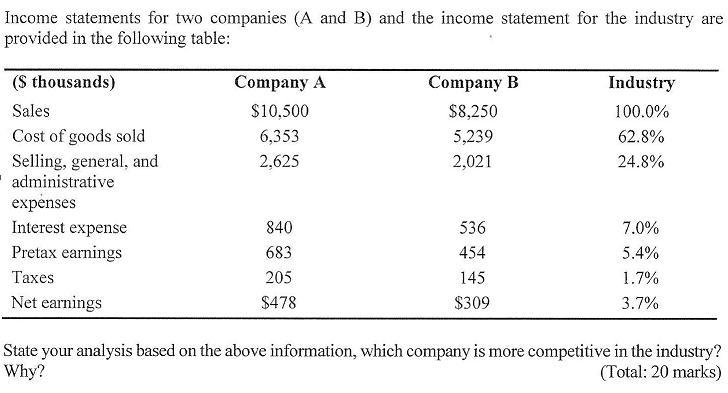
Income statements for two companies (A and B) and the income statement for the industry are...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Income statements for two companies (A and B) and the income statement for the industry are provided in the following table: (S thousands) Sales Cost of goods sold Selling, general, and administrative expenses Interest expense Pretax earnings Taxes Net earnings Company A $10.500 6,353 2,625 840 683 205 $478 Company B $8,250 5,239 2,021 536 454 145 $309 Industry 100.0% 62.8% 24.8% 7.0% 5.4% 1.7% 3.7% State your analysis based on the above information, which company is more competitive in the industry? Why? (Total: 20 marks) Income statements for two companies (A and B) and the income statement for the industry are provided in the following table: (S thousands) Sales Cost of goods sold Selling, general, and administrative expenses Interest expense Pretax earnings Taxes Net earnings Company A $10.500 6,353 2,625 840 683 205 $478 Company B $8,250 5,239 2,021 536 454 145 $309 Industry 100.0% 62.8% 24.8% 7.0% 5.4% 1.7% 3.7% State your analysis based on the above information, which company is more competitive in the industry? Why? (Total: 20 marks)
Expert Answer:
Answer rating: 100% (QA)
We can examine several financial measures and compare them to industry averages to ascertain which c... View the full answer

Related Book For 

Fundamentals of Analytical Chemistry
ISBN: 978-0495558286
9th edition
Authors: Douglas A. Skoog, Donald M. West, F. James Holler, Stanley R. Crouch
Posted Date:
Students also viewed these chemistry questions
-
A solution was prepared by dissolving 5.76 g of KCl MgCl2 6H2O (277.85 g/mol) in sufficient water to give 2.000 L. Calculate (a) The molar analytical concentration of KCl MgCl2 in this solution....
-
Solution A was prepared by dissolving potassium acetate in methanol. Solution B was prepared by adding potassium methoxide to acetic acid. Reaction of methyl iodide either with solution A or with...
-
A solution was prepared by dissolving 0.834 g of sulfur, S8, in 100.0 g of acetic acid, HC2H3O2. Calculate the freezing point and boiling point of the solution.
-
Thor Bhd. (Thor) is a listed company in Malaysia, specializes in selling batteries. At 31 December 2021, Thor holds four distinct types of batteries in its warehouse. The accountant of Thor provided...
-
The Kenyon Manufacturing Company is interested in selecting the best location for a new plant. After a detailed study of 10 sites, the three location finalists are Georgetown, Kentucky; Marysville,...
-
At what brick-and-mortar store did you have your worst experience? Describe this store.
-
A Heartland Monitor poll taken in December 2016 obtained responses from 990 adult Americans to the question How would you rate the current state of the economy? Is it excellent, good, fair, or poor?...
-
Friedman Construction reported taxable income of $50,000 for 2011, its first fiscal year. The enacted tax rate for 2011 is 40%. Enacted tax rates and deductible amounts for 20122014 are as follows:...
-
If the Bank of Canada sells $1 million of bonds to Irving the Investor, who pays for the bonds with a briefcase filled with currency, what happens to reserves and the monetary base? First show the...
-
A solar collector design consists of several parallel tubes each enclosed concentrically in an outer tube which is transparent to solar radiation. The tubes are thin walled with diameter of the inner...
-
Find the interest rate for a principal of $6463 and charged $47348 in interest for 11 years.
-
How do we establish a relationship between Strategic Directions and Vision of a company .
-
Doorman Sportswear's articles of incorporation authorize the company to issue 6,000 $7 preferred shares and 850,000 common shares. Doorman issued 1,200 preferred shares at $100 per share. It issued...
-
Suppose a 250. mL flask is filled with 0.60 mol of NO2 and 0.80 mol of O2. This reaction becomes possible: 2NO2(g) 2NO(g) + O2(g) Complete the table below, so that it lists the initial molarity of...
-
Give a tight asymptotic run time analysis ( notation) for the following pseudo-code snippet: L-new linked list containing the values (1, 2, ..., n) for i -1 to n do p - L.head for j 0 to i-1 do...
-
Suppose that a company decides to raise capital by selling stock. Over the next 10 years the average monthly price of the stock fluctuates according to the rule S(t) = 0.17t5-2.04t+ 65.15 where S(t)...
-
Harold is responsible for reporting on the performance of a portfolio which consists of investments in two bonds, A and B. Bond A matures in two years time, has a coupon rate of 10% pa, a par value...
-
The bookkeeper for Riley, Inc., made the following errors: a. A cash purchase of supplies of $357 was recorded as a debit to Supplies for $375 and a credit to Cash of $375. b. A cash sale of $3,154...
-
Supply the missing data in the table below. Molar Analytical Concentration, Cr Lactic Iodic Butanoic Hypochlorous 0.280 7.00 Nitrous Hydrogen .120 _ 0.200 , 0.640 0.765 5.00 0.644_ __ _0.105 0.413...
-
Calculate the wavelength of the sodium line at 589 nm in an aqueous solution with a refractive index of 1.35.
-
Calculate the pH of a solution that is (a) 0.0100 M in HClO4 and 0.0300 M in monochloroacetic acid. (b) 0.0100 M in HCl and 0.0150 M in H2SO4. (c) 0.0100 M in NaOH and 0.0300 M in Na2S. (d) 0.0100 M...
-
Why does a confidence interval contain more information than the result of a single hypothesis test?
-
Let \(Y\) be a Bernoulli random variable with success probability \(\operatorname{Pr}(Y=1)=p\), and let \(Y_{1}, \ldots, Y_{n}\) be i.i.d. draws from this distribution. Let \(\hat{p}\) be the...
-
Sketch a hypothetical scatterplot for a sample of size 10 for two random variables with a population correlation of (a) 1.0; (b) -1.0 ; (c) 0.9; (d) -0.5 ; (e) 0.0.

Study smarter with the SolutionInn App


