Specify the hydrogen bonding behavior of the 4 species below by selecting: donor for species that...
Fantastic news! We've Found the answer you've been seeking!
Question:
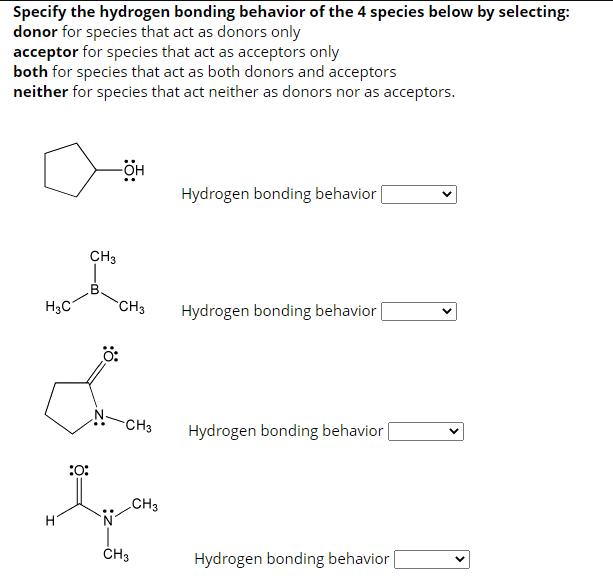
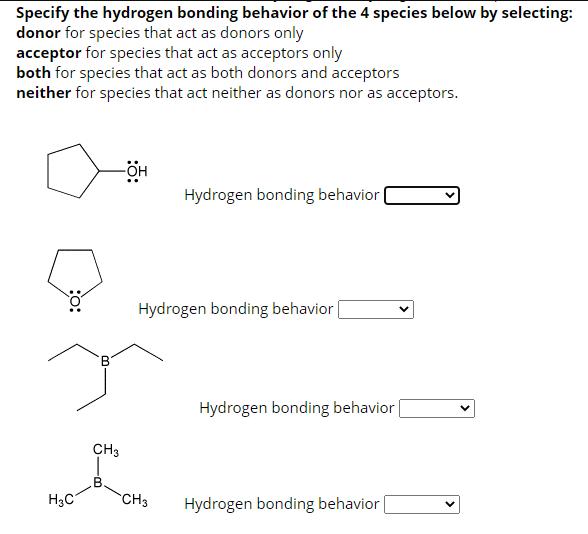
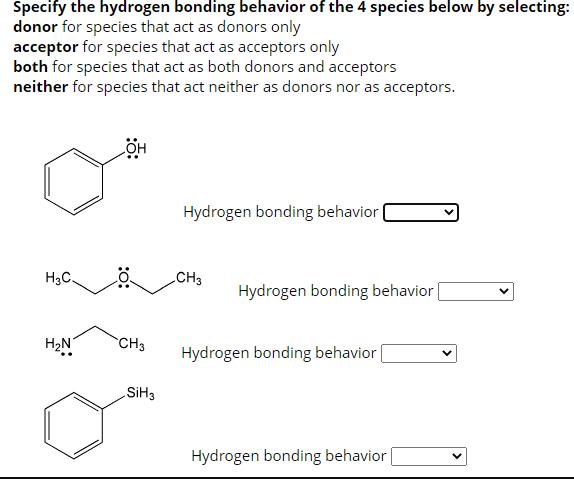
Transcribed Image Text:
Specify the hydrogen bonding behavior of the 4 species below by selecting: donor for species that act as donors only acceptor for species that act as acceptors only both for species that act as both donors and acceptors neither for species that act neither as donors nor as acceptors. H3C -OH CH3 CH3 Hydrogen bonding behavior 0: L -CH3 Hydrogen bonding behavior :0: ton CH3 H CH3 Hydrogen bonding behavior Hydrogen bonding behavior Specify the hydrogen bonding behavior of the 4 species below by selecting: donor for species that act as donors only acceptor for species that act as acceptors only both for species that act as both donors and acceptors neither for species that act neither as donors nor as acceptors. :0: H₂C B -OH CH3 Hydrogen bonding behavior | Hydrogen bonding behavior CH3 Hydrogen bonding behavior [ Hydrogen bonding behavior Specify the hydrogen bonding behavior of the 4 species below by selecting: donor for species that act as donors only acceptor for species that act as acceptors only both for species that act as both donors and acceptors neither for species that act neither as donors nor as acceptors. H3C. H₂N CH3 SiH3 Hydrogen bonding behavior CH3 Hydrogen bonding behavior Hydrogen bonding behavior | Hydrogen bonding behavior | Specify the hydrogen bonding behavior of the 4 species below by selecting: donor for species that act as donors only acceptor for species that act as acceptors only both for species that act as both donors and acceptors neither for species that act neither as donors nor as acceptors. H3C -OH CH3 CH3 Hydrogen bonding behavior 0: L -CH3 Hydrogen bonding behavior :0: ton CH3 H CH3 Hydrogen bonding behavior Hydrogen bonding behavior Specify the hydrogen bonding behavior of the 4 species below by selecting: donor for species that act as donors only acceptor for species that act as acceptors only both for species that act as both donors and acceptors neither for species that act neither as donors nor as acceptors. :0: H₂C B -OH CH3 Hydrogen bonding behavior | Hydrogen bonding behavior CH3 Hydrogen bonding behavior [ Hydrogen bonding behavior Specify the hydrogen bonding behavior of the 4 species below by selecting: donor for species that act as donors only acceptor for species that act as acceptors only both for species that act as both donors and acceptors neither for species that act neither as donors nor as acceptors. H3C. H₂N CH3 SiH3 Hydrogen bonding behavior CH3 Hydrogen bonding behavior Hydrogen bonding behavior | Hydrogen bonding behavior |
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these psychology questions
-
Q.4. Solve the following problem using the following data: Task Predecessor ta tm tb A 1 9. 2 5 8. C 1 3 ---- D A 4 10 25 E A 3 7 12 F 10 15 25 G C 9 14 H D,E 7 I D,E, F 1 6. J D, E, F, G 2 10 K H,...
-
How do the psychological phenomena of loss aversion and the endowment effect influence stakeholder perceptions and attitudes towards impending organizational change initiatives, and what mechanisms...
-
How do advanced organizations cultivate a conducive ecosystem that fosters creativity and innovation, leveraging sophisticated methodologies to curate dynamic work environments that stimulate...
-
Adrian Webb bought 618 bricks to build a barbecue pit, paying $185.40. Find the cost per brick. (HINT: Cost per brick means the cost for one brick.) The cost per brick is s (Round to nearest hundidth)
-
A 2-L liquid is mixed with 3 L of another liquid, forming a homogeneous liquid solution at the same temperature and pressure. Can the volume of the solution be more or less than the 5 L? Explain.
-
Solve each equation in Exercises 6870 using the quadratic formula. 2x = 3 - 4x
-
What are the key factors behind a successful report presentation?
-
The normal capacity of a manufacturing plant is 30,000 direct labor hours or 20,000 units per month. Standard fixed costs are $6,000, and variable costs are $12,000. Data for two months follow: For...
-
Adhering to social, business and ethical standards is important for a business to both develop and secure positive referrer relationships. For a Finance Broker, these standards could be acting in the...
-
The COVID-19 pandemic affected companies in the food industry in unique ways, particularly during 2020. Ruth's Hospitality Group (Ticker: RUTH) develops and operates fine dining restaurants under the...
-
You are a security professional. You have been asked to develop strategies to protect data and file systems in a small Linux environment. The environment contains a Linux application server, a Linux...
-
What is the relationship between communication and health inequality? Specify the pathways through which communication influences health inequality.
-
Prisons for women differ from male facilities in many ways. They are less violent, women maintain closer ties with staff, and their facilities are not infested with gangs. It is also the case that...
-
Read the scenario below and answer ALL the questions that follow Company X is a well-established company operating in South Africa, known for its diverse workforce. However, it has a company policy...
-
Please answer the below questions in 250-500 words (all together). Please use only the Week 6 reading/video assignment (Chapter 1 of Democracy: A Very Short Introduction and Dr. Kietlinski's recorded...
-
In 500 words or more for each question, please answer the following questions. Do you agree with the philosophy of technological realism? what definition would you share with a layman? Explain the...
-
Find the absolute extrems of the function on the closed interval. Use a graphing utility to verify your results. (If an answer does not exist, enter DN f(x)=x-6x-2, [0, 3] absolute maximum (x, y) =...
-
Find the equation of the plane passing through the points P 5,4,3 ,Q 4,3,1 and R 1,5,4
-
Diethyl ether has a boiling point of 34.5°C, and 1-butanol has a boiling point of 117°C: Both of these compounds have the same numbers and types of atoms. Explain the difference in their...
-
Write equations representing the following processes: (a) The electron affinity of S-. (b) The third ionization energy of titanium. (c) The electron affinity of Mg2+. (d) The ionization energy of O2-.
-
A silver rod and a SHE are dipped into a saturated aqueous solution of silver oxalate, Ag2C2O4, at 25C. The measured potential difference between the rod and the SHE is 0.589 V, the rod being...
-
For the periodic processes shown below: a. Schedule the processes using an RMS policy. b. Schedule the processes using an EDF policy. In each case, compute the schedule for an interval equal to the...
-
For the periodic processes shown below: a. Schedule the processes using an RMS policy. b. Schedule the processes using an EDF policy. In each case, compute the schedule for an interval equal to the...
-
For the given periodic process execution times and periods (P1 has the highest priority), show how much CPU time of higher-priority processes will be required during one period of each of the...

Study smarter with the SolutionInn App


