(a) Draw both the ac equivalent circuit and small-signal equivalent circuit of the configuration in Figure...
Fantastic news! We've Found the answer you've been seeking!
Question:
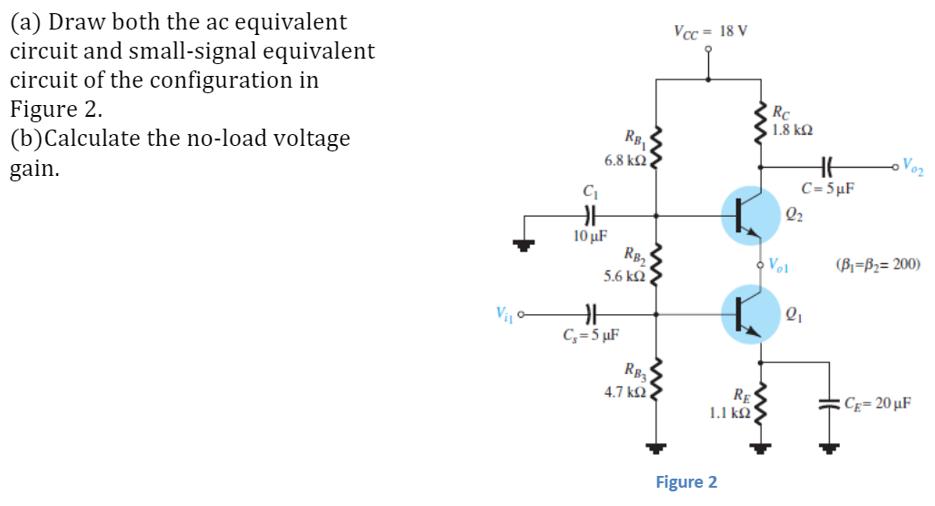
Transcribed Image Text:
(a) Draw both the ac equivalent circuit and small-signal equivalent circuit of the configuration in Figure 2. (b)Calculate the no-load voltage gain. V₁₁ RB₁ 6.8 ΚΩ , C₁ HE 10 μF RB₂ 5.6 ΚΩ | C₁=5 μF RB3 4,7 ΚΩ , Vcc= 18 V K Figure 2 RE 1.1 ΚΩ Rc 1.8 ΚΩ HH C=5µF 2₂ Vol K 2₁ Voz (B₁-B₂= 200) CE= 20 µF (a) Draw both the ac equivalent circuit and small-signal equivalent circuit of the configuration in Figure 2. (b)Calculate the no-load voltage gain. V₁₁ RB₁ 6.8 ΚΩ , C₁ HE 10 μF RB₂ 5.6 ΚΩ | C₁=5 μF RB3 4,7 ΚΩ , Vcc= 18 V K Figure 2 RE 1.1 ΚΩ Rc 1.8 ΚΩ HH C=5µF 2₂ Vol K 2₁ Voz (B₁-B₂= 200) CE= 20 µF
Expert Answer:
Answer rating: 100% (QA)
Image sent Image of the AC equivalent circuit of the configuration in Figure 2 Answer The AC equival... View the full answer

Related Book For 

Managerial Accounting
ISBN: 978-0078111006
14th edition
Authors: Ray Garrison, Eric Noreen and Peter Brewer
Posted Date:
Students also viewed these electrical engineering questions
-
Prove that the Chebyshev polynomials of the first kind can be expressed as Tn(x) Tn(x) = cos(narccos(x)) 4 4
-
a) Plot the function y = 3x-6x+x-2 from = -20 to x = 20. 2+3+2 b) What is the long term behavior of the function, as a approaches oo? What is the long term behavior as approaches-co? Select the...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
The mass-to-charge ratio for the positive ion F+ is 1.97 107 kg/C. Using the value of 1.602 1019 C for the charge on the ion, calculate the mass of the fluorine atom. (The mass of the electron is...
-
A thermocouple is used to measure the temperature of a flame in a combustion chamber. If the thermocouple temperature is 1033 K and the walls of the chamber are at 700 K, what is the error in the...
-
For the following exercises, use the graphs to write a polynomial function of least degree. (-300,0) f(x) 2-10 1-10 (100, 0) 30-200 -100 100 200 -1-10(0, -90,000) -2.16 10 -4.10
-
The compressibility factor \(Z\) of an ideal gas is always (a) 0 (b) 1 (c) \(>1\) (d) \( <1\).
-
Xiong Co. uses a periodic inventory system. Its records show the following for the month of May, in which 65 units were sold. Instructions Compute the ending inventory at May 31 and cost of goods...
-
Find the solution of the exponential equation, correct to four decimal places. e-3x=9 x=
-
A dam is to be constructed using the cross-section shown. Assume the dam width is w = 160 ft. For water height H = 9 ft. calculate the magnitude and line of action of the vertical force of water on...
-
You are a CEO at a finance firm. One of your managers is up for promotion. You want to make sure that they can discern a good financial offer from a bad one so that they will do well making the...
-
Calculate the life insurance gross premium for an insurance company, XYZ Life, under the following assumptions. Please round your answer to the nearest cent. 1-year term insurance for a 30-year-old...
-
In Government Contracting what are improper Terminations for Default and are they converted to Terminations for convenience always, sometimes, or never?
-
Some consider workplace bullying as behavior comparable to harassment based on sex or some other protected characteristic, but despite attempts, no laws have been passed to protect victims from...
-
Analyze whether you believe the Dobbs case was correctly decided and discuss which of the opinions in the Dobbs case (majority, concurring, dissenting) is correct in your personal opinion and why. Be...
-
Case - Smith v. Delta Tau Delta, Inc. - 9 N.E.3d 154 (2014) - Supreme Court of Indiana Issue - Is there an agency relationship between the local fraternity and its members and the national...
-
Joseph and Margaret recently had their first child. They're excited about building their family together. Margaret wants to begin a college savings fund for their child immediately, whereas Joseph...
-
Nate prepares slides for his microscope. In 1 day he prepared 12 different slides. Which equation best represents y, the total number of slides Nate prepares in x days if he continues at this rate? A...
-
For the just completed year, Strident Company had net income of $84,000. Balances in the companys current asset and current liability accounts at the beginning and end of the year were as follows:...
-
Annual cash inflows from two competing investment opportunities are given below. Each investment opportunity will require the same initial investment. Required: Compute the present value of the cash...
-
The marketing department of Graber Corporation has submitted the following sales forecast for the upcoming fiscal year. The selling price of the companys product is $22.00 per unit. Management...
-
What would the QA/QC planning grid (see Figure 12. 6) look like for a project you have been involved in? Data from Figure 12. 6 Table 12.6 QA/QC planning grid, with examples Quality assurance...
-
Why is the definition of quality critical to the success and acceptance of a project?
-
What is the purpose of the Quality Management Plan?

Study smarter with the SolutionInn App


