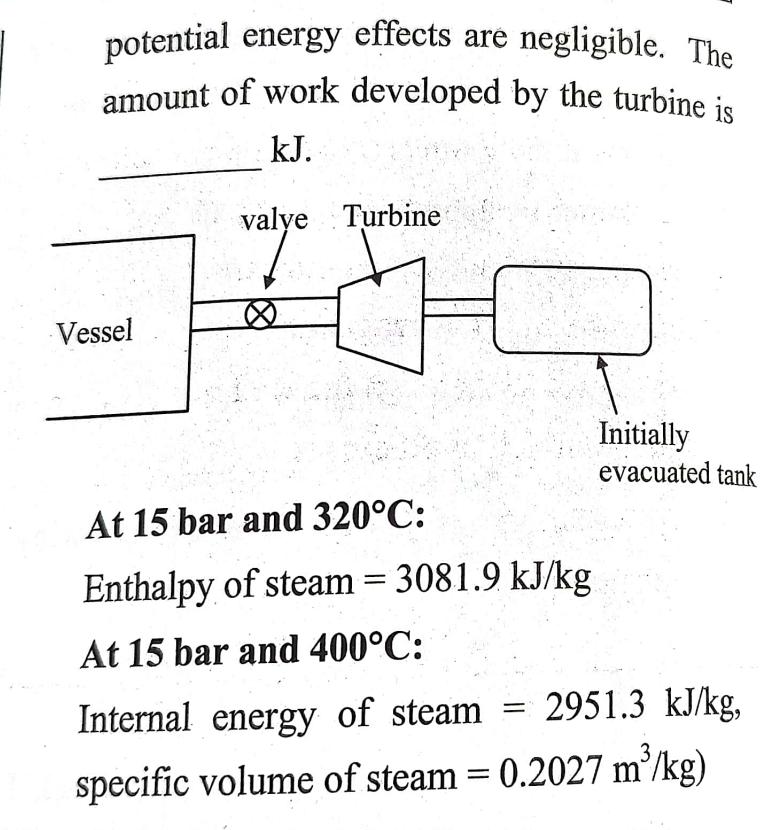
Steam at a pressure of 15 bar and a temperature of 320C is contained in a...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
Steam at a pressure of 15 bar and a temperature of 320°C is contained in a large vessel shown below. Connected to the vessel through a valve is a turbine followed by a small initially evacuated tank with a volume of 0.6 m³. When power is required, the valve is opened and the tank fills with steam until the pressure is 15 bar. The temperature in the tank is then 400°C. The filling process takes place adiabatically and kinetic and 3 potential energy effects are negligible. The amount of work developed by the turbine is kJ. Vessel valve Turbine valye 70 X At 15 bar and 320°C: Enthalpy of steam = 3081.9 kJ/kg At 15 bar and 400°C: = Initially evacuated tank Internal energy of steam 2951.3 kJ/kg, 3 specific volume of steam = 0.2027 m²/kg) Steam at a pressure of 15 bar and a temperature of 320°C is contained in a large vessel shown below. Connected to the vessel through a valve is a turbine followed by a small initially evacuated tank with a volume of 0.6 m³. When power is required, the valve is opened and the tank fills with steam until the pressure is 15 bar. The temperature in the tank is then 400°C. The filling process takes place adiabatically and kinetic and 3 potential energy effects are negligible. The amount of work developed by the turbine is kJ. Vessel valve Turbine valye 70 X At 15 bar and 320°C: Enthalpy of steam = 3081.9 kJ/kg At 15 bar and 400°C: = Initially evacuated tank Internal energy of steam 2951.3 kJ/kg, 3 specific volume of steam = 0.2027 m²/kg)
Expert Answer:
Related Book For 

Thermodynamics An Interactive Approach
ISBN: 978-0130351173
1st edition
Authors: Subrata Bhattacharjee
Posted Date:
Students also viewed these mechanical engineering questions
-
A long tube contains air at a pressure of 1.00 atm and a temperature of 77.0C. The tube is open at one end and closed at the other by a movable piston. A tuning fork near the open end is vibrating...
-
A large supply line carries water at a pressure of 1 MPa. A small leak with an area of 1 cm2 develops on the pipe which is exposed to the atmosphere at 100 kPa. Assuming the resulting flow to be...
-
A cylinder contains oxygen at a pressure of 2.00 atm. The volume is 4.00 L. and the temperature is 300 K. Assume that the oxygen may be treated as an ideal gas. The oxygen is carried through the...
-
Following is the condensed balance sheet of Martinez, O'Neill and Clemens, partners who share profits or losses in the ratio of 2 : 3 : 5. Cash $50,000 Liabilities $200,000 Other assets 1,050,000...
-
What political and religious beliefs and institutions gave Hebrew civilization its unique character and account for its important legacy to Western civilization?
-
The arc AB of a circle, center O and radius x cm, is such that angle AOB = /12 radians. Given that the arc length AB is l cm, a. Show that the area of the sector can be written as -6l 2 / b. Find the...
-
A flywheel: (a) Is provided to minimize the engine vibration (b) Is provided to control engine speed (c) Controls output fluctuation and input accordingly (d) All of the above
-
Cooper Construction is considering purchasing new, technologically advanced equipment. The equipment will cost $625,000 with a salvage value of $50,000 at the end of its useful life of 10 years. The...
-
What is the profit under the "best case scenario? According to the scenario manager (output), what is the profit under the "most likely" scenario? According to the one variable DATA TABLE, what is...
-
Using the information in Table 7.1, construct another table showing how a lump-sum franchise tax of $30 affects the various average cost curves of the firm. Table 7.1 Average Variable Cost, AVC =...
-
Adam Bedwell died in 2012, leaving an estate of $27,000,000. Adam's wife died in 2009. In 2009, Adam gave his son property that resulted in a taxable gift of $6,000,000 and upon which Adam paid...
-
TCP is a very symmetric protocol, but the client/server model is not. Consider an asymmetric TCP-like protocol in which only the server side is assigned a port number visible to the application...
-
An organization has been assigned the prefix 200.1.1/24 (a class C) and wants to form subnets for four departments, with hosts as follows: There are 145 hosts in all. (a) Give a possible arrangement...
-
Learn about a key escrow, or key surrender, scheme (for example, Clipper). What are the pros and cons of key escrow?
-
In stop-and-wait transmission, suppose that both sender and receiver retransmit their last frame immediately on receipt of a duplicate ACK or data frame; such a strategy is superficially reasonable...
-
You are an Internet service provider; your client hosts connect directly to your routers. You know some hosts are using experimental TCPs and suspect some may be using a greedy TCP with no congestion...
-
10-yr bond, par value = $1000 Selling price = $1,134.20 10.0% annual coupon rate Callable after 4 years What is the bonds Yield to Call (YTC)?
-
What is the shape of the exponential distribution?
-
Octane (C8H18) is burned with theoretical amount of air at a pressure of 500 kPa. Determine (a) The air fuel ratio on a mole basis. (b) The air fuel ratio on a mass basis. (c) If the products are...
-
Hydrogen is produced at 100 kPa and 298 K from a mixture of hydrogen and methane containing 10% hydrogen and 90% methane by volume. If the device has an energetic efficiency of 20%. Determine the...
-
A 40 kg aluminum block at 90oC is dropped into an insulated tank that contains 0.5 m3 of liquid water at 20oC. Determine the equilibrium temperature.
-
What connection did Newton make between a falling apple and the Moon?
-
How was Pioneer 10 able to escape the solar system with an initial speed less than escape speed?
-
If you stood atop a super-tall ladder three times as far from Earths center as at Earths surface, how would your weight compare with it present value?

Study smarter with the SolutionInn App



