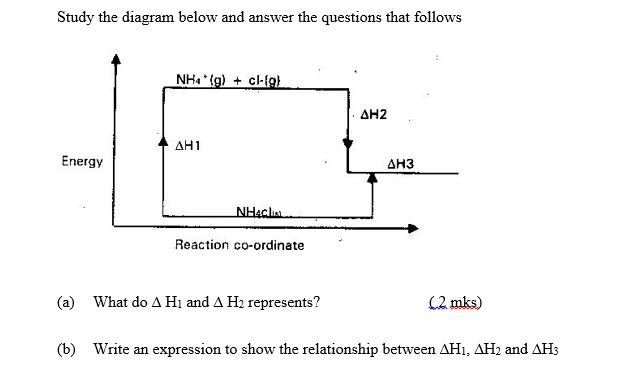
Study the diagram below and answer the questions that follows Energy NH4(g) + cl-(g) AH1 NH4Cl...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Study the diagram below and answer the questions that follows Energy NH4(g) + cl-(g) AH1 NH4Cl Reaction co-ordinate (a) What do A H₁ and A H₂ represents? ΔΗ2 ΔΗ3 (2. mks) (b) Write an expression to show the relationship between AH1, AH2 and AH3 Study the diagram below and answer the questions that follows Energy NH4(g) + cl-(g) AH1 NH4Cl Reaction co-ordinate (a) What do A H₁ and A H₂ represents? ΔΗ2 ΔΗ3 (2. mks) (b) Write an expression to show the relationship between AH1, AH2 and AH3
Expert Answer:
Answer rating: 100% (QA)
a Here H1 and H2 represent the enthalpies of reaction 1 and reaction 2 respectively Enthalpy is r... View the full answer

Related Book For 

Income Tax Fundamentals 2013
ISBN: 9781285586618
31st Edition
Authors: Gerald E. Whittenburg, Martha Altus Buller, Steven L Gill
Posted Date:
Students also viewed these chemistry questions
-
Study the article below and answer the questions that follow: Drug firm Adcock stays in SA hands Johannesburg -Chile's CFR Pharmaceuticals said on Friday it would drop its $1.2bn bid for drugmaker...
-
Study the article below and answer the questions that follow. Panasonic Scouts U.S. Sites for New Tesla Battery Factory Company eyes locations in Oklahoma, Kansas; talks ongoing Plant to turn out...
-
Study the information given below and answer the questions that follow. Element Atomic radius(nm) Ionic radius nm Formula of oxide Melting point( 0 c) A 0.364 0.421 A 2 O -119 D 0.830 0.711 D O 2 837...
-
A line charge density pL of length L lies parallel to an infinite sheet of surface charge density ps. How much work is required to rotate the line charge so that it is vertical?
-
RKR Dance Studios hired Jessica Makowski as an at-will employee. At that time, Makowski signed a noncompete agreement. In 2006, Makowski signed a new noncompete, allegedly in consideration for new...
-
Presented next are the ending balances of accounts for the Kansas Instruments Corporation at December 31, 2021. Additional Information:1. The notes receivable, along with any accrued interest, are...
-
How do sequence diagrams for design differ from those for analysis? In which one do boundary objects appear? Why?
-
How does factor analysis allow for data reduction?
-
A Describe the relationship between a and b that will make the value of the expression 7x equal to 7.
-
Prepare journal entries for the above transactions. Aylmer Industries Inc. (Aylmer) has been in business since 2018. Aylmer has two main business segments. This first segment is the construction of...
-
Mandoza Mines was established 10 years ago as an affiliate of Polyus Gold, a Russian-based gold mining company. Mandoza mines was affected severely in the global financial crisis of 2007 and 2008 and...
-
What is a possible output of the following application? A. Your gift: wrap.Gift@29ca2745 B. Your gift: Your gift: C. It does not compile. D. It compiles but throws an exception at runtime. package...
-
What is the output of the following application? A. Walking and running! B. Walking and jogging! C. Sprinting! D. The code does not compile. E. The code compiles but prints an exception at runtime....
-
What is the command to call one constructor from another constructor in the same class? A. construct() B. parent() C. super() D. this() E. that()
-
Which can implement a functional interface? A. An anonymous class B. A top-level class C. A lambda expression D. An anonymous class or a top-level class E. A top-level class or a lambda expression F....
-
What is the result of executing the Grasshopper program? A. The code prints hop once. B. The code prints hop twice. C. The first compiler error is on line p1. D. The first compiler error is on line...
-
Earl Lee Riser Alarm Co. issued $10,000 of bonds on January 1, 2021. The bonds pay interest semiannually. This is a partial bond amortization schedule for the bonds. Effective interest Decrease in...
-
The vapor pressure of the liquid NH, is measured at different temperatures. The following vapor pressure data are obtained. Temperature, K P, mmHg 217.1 223.4 234.7 588.1 Calculate the enthalpy of...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family. Ivan and Irene have the following investment income, in addition to that reported in Chapter 1: Dividends...
-
Tom has a successful business with $100,000 of income in 2012. He purchases one new asset in 2012, a new machine which is 7-year MACRS property and costs $25,000. If you are Tom's tax advisor, how...
-
Leslie is a single taxpayer who is under age 65 and in good health. For 2012, she has a salary of $23,000 and itemized deductions of $1,000. Leslie is entitled to one exemption on her tax return. a....
-
If a politician running for office does not speak in general terms, does not try to move to the middle of the political spectrum, and does not take polls, is the median voter model therefore wrong?
-
Voters often criticize politicians running for office who do not speak in specific terms (i.e., who do not specify which spending programs will be cut, whose taxes will be raised, etc.). If voters...
-
Why is rent-seeking activity socially wasteful?

Study smarter with the SolutionInn App


