Sulphur burns in air to form sulphur (IV) oxide. A simple energy level diagram for the...
Fantastic news! We've Found the answer you've been seeking!
Question:
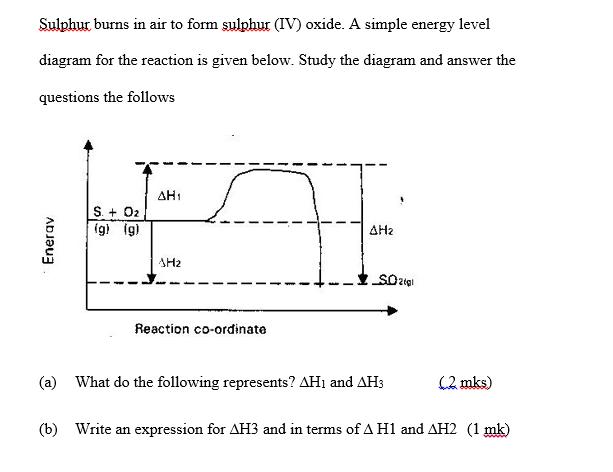
Transcribed Image Text:
Sulphur burns in air to form sulphur (IV) oxide. A simple energy level diagram for the reaction is given below. Study the diagram and answer the questions the follows Enerav S. + O2 (g) (g) AH₁ SH₂ Reaction co-ordinate 1 1 AH₂ SO2(gl (a) What do the following represents? AH1 and AH3 (2 mks) (b) Write an expression for AH3 and in terms of A H1 and AH2 (1 mk) Sulphur burns in air to form sulphur (IV) oxide. A simple energy level diagram for the reaction is given below. Study the diagram and answer the questions the follows Enerav S. + O2 (g) (g) AH₁ SH₂ Reaction co-ordinate 1 1 AH₂ SO2(gl (a) What do the following represents? AH1 and AH3 (2 mks) (b) Write an expression for AH3 and in terms of A H1 and AH2 (1 mk)
Expert Answer:
Answer rating: 100% (QA)
AH is the activation for the reaction energy Refers to the energy ... View the full answer

Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Below is the energy level diagram for the reaction H 2(g) + F 2(g) HF (g) (a) Calculate the heat of formation of HF (g) ( 2 msk) (b) Is this reaction exothermic or endothermic? Enerav (KJ) 411 389...
-
Given the following reaction coordinate diagram for the reaction of A to give D, answer the following questions: a. How many intermediates are there in the reaction? b. How many transition states are...
-
For each reaction, estimate whether ÎSo for the reaction is positive, negative, or impossible to predict. a. (b) The formation of diacetone alcohol: c. heat C10H22--> n-decane C3H6 + C7H16...
-
Reynolds number is: i. a dimensionless quantity ii. inversely proportional to viscosity of liquid iii. directly proportional to velocity of liquid iv. used for quantifying the laminar or turbulent...
-
Communication Technical Systems, Inc., (CTS) began providing computer programming services for Gateway 2000, Inc., (Gateway), in July 1994. Rickey Densmore, a programmer for CTS, worked on the...
-
The 2021 balance sheet for Hallbrook Industries, Inc., is shown below. The company?s 2021 income statement reported the following amounts ($ in thousands): Net sales...
-
Explain the three types of visibility for package elements: public, private, and protected.
-
Jason, Kelly, and Becky, who share partnership profits 50 percent, 30 percent, and 20 percent, respectively, decide to liquidate their partnership. They need the cash from the partnership as soon as...
-
4. Show that if z = i and z = i - 1, then Log 2122 Log 21 + Log 22
-
Positive charges Q and Q' are placed on opposite sides of a grounded sphere of radius R at distances of 2R and 4R, respectively, from the sphere center. Show that Q' is repelled from the sphere if Q'...
-
A company wants to analyze this investment: initial investmentof $80.000.000, $30.000 of income in the first year then 10% increase over previous year's revenue for 5 years, residual value of $20.000...
-
What does the following print? A. false, false, false B. false, false, true C. false, true, true D. true, false, true E. true, true, false F. true, true, true interface Vehicle {} class Bus...
-
Imagine you are working with another team to build an application. You are developing code that uses a class that the other team has not finished writing yet. You want to allow easy integration once...
-
What is the output of the following application? A. Honk(1)! B. Honk(3)! C. The code does not compile. D. The code compiles, but the output cannot be determined until runtime. E. None of the above....
-
What is the output of the InfiniteMath program? A. 2.0 B. 4.0 C. 8.0 D. The code does not compile. E. The code compiles but prints an exception at runtime. F. None of the above. class Math { public...
-
Which of the following are the best reasons for creating a default interface method? (Choose two.) A. Allow interface methods to be overloaded. B. Add backward compatibility to existing interfaces....
-
Consider the hypothetical economies of Hestiatia and Vanaheim, both of which produce bottles of bulg using only workers and tools. Suppose that, during the course of 35 years, the level of physical...
-
What are three disadvantages of using the direct write-off method?
-
A shampoo solution at 25C has a hydroxide-ion concentration of 1.5 10-9 M. Is the solution acidic, neutral, or basic?
-
An electrochemical cell is made by placing a zinc electrode in 1.00 L of 0.200 M ZnSO4 solution and a copper electrode in 1.00 L of 0.0100 M CuCl2 solution. a. What is the initial voltage of this...
-
A 25.00-mL sample contains 0.562 g of NaHCO3. This sample is used to standardize an NaOH solution. At the equivalence point, 42.36 mL of NaOH has been added. a. What was the concentration of the...
-
Victoria has prepared the following list of statements about corporations. 1. A corporation is an entity separate and distinct from its owners. 2. As a legal entity, a corporation has most of the...
-
Indicate whether the following items would appear on the income statement (IS), statement of financial position (SFP), or retained earnings statement (RES). a. Dividends. b. Cash. c. Salaries and...
-
Victoria has studied the information you gave her in that exercise and has come to you with more statements about corporations. 1. Corporation management is both an advantage and a disadvantage of a...

Study smarter with the SolutionInn App


