Suppose 200 mol/s of liquid methanol and 30% excess oxygen are fed to a reactor at...
Fantastic news! We've Found the answer you've been seeking!
Question:
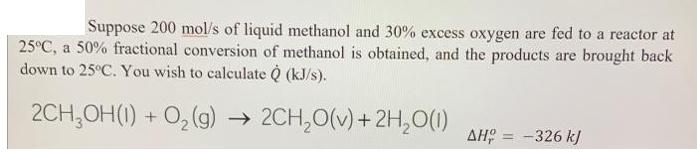
Transcribed Image Text:
Suppose 200 mol/s of liquid methanol and 30% excess oxygen are fed to a reactor at 25°C, a 50% fractional conversion of methanol is obtained, and the products are brought back down to 25°C. You wish to calculate Q (kJ/s). 2CH₂OH(1) + O₂(g) → 2CH₂O(v) + 2H₂O(1) ΔΗ, = -326 kJ Suppose 200 mol/s of liquid methanol and 30% excess oxygen are fed to a reactor at 25°C, a 50% fractional conversion of methanol is obtained, and the products are brought back down to 25°C. You wish to calculate Q (kJ/s). 2CH₂OH(1) + O₂(g) → 2CH₂O(v) + 2H₂O(1) ΔΗ, = -326 kJ
Expert Answer:
Answer rating: 100% (QA)
The reaction involved is 2CH3OH302 2CO24H20 Thus feed has 20... View the full answer

Related Book For 

Elementary Principles of Chemical Processes
ISBN: 978-0471720638
3rd Edition
Authors: Richard M. Felder, Ronald W. Rousseau
Posted Date:
Students also viewed these accounting questions
-
One gram-mole each of CO 2 , O 2 , and N 2 are fed to a batch reactor and heated to 3000K and 5.0atm. The two reactions given here proceed to equilibrium (also shown are the equilibrium constants at...
-
A cylinder fitted with a piston contains liquid methanol at 20C, 100 kPa and volume 10 L. The piston is moved, compressing the methanol to 20 MPa at constant temperature. Calculate the work...
-
A cylinder fitted with a piston contains liquid methanol at 20C, 100 kPa and volume 10 L. The piston is moved, compressing the methanol to 20 MPa at constant temperature. Calculate the work required...
-
A Hydrogen atom has one proton in the nucleus and one electron in the shell. In a classic model of the atom, in a certain state, this electron is in a circular orbit around the nucleus with an...
-
Explain relationships between the corporate opportunity doctrine and the business judgment rule.
-
What does the abbreviation OAR stand for?
-
A ball falling near the surface of the Earth where air friction is not negligible eventually slows down to a constant "terminal" velocity. How is that possible given that frictional force increases...
-
The Ewert Exploration Company is considering two mutually exclusive plans for extracting oil on property for which it has mineral rights. Both plans call for the expenditure of $10 million to drill...
-
A multinational corporation is accused of environmental pollution and human rights violations in a foreign country where it operates. What legal challenges arise in holding the corporation...
-
Using the alternative-parameter method, determine the parameters of the following distributions based on the given assessments. Refer to Step 5.5 if necessary. a. Find the parameter value for the...
-
The taxpayer is engaged in business with the following data for taxable year 2021, the first year of operation: MANILA Sales Rent income Cash dividends from foreign corporation Property dividends...
-
Define politics and explain how politics differs from power.
-
Which of the following is not a PFP service? A. Designing financial strategies. B. Identifying personal financial goals and resources. C. Making personalized recommendations. D. Preparing individual...
-
Write a static method max3() that takes three int arguments and returns the value of the largest one. Add an overloaded function that does the same thing with three double values.
-
Who are the commonly reported victims of female serial killers? What are some of the motivations of female serial killers?
-
The members evaluation of progress toward achieving the clients PFP goals is an element of which type of engagement? A. PFP engagement. B. Implementation engagement. C. Monitoring engagement. D....
-
Make balance sheet given the following information for Corp. as of December 31, 2014: cash = $200,000; intangible net fixed assets = $854,000; accounts payable = $289,000; accounts receivable =...
-
PC Contractors, Inc., was an excavating business in Kansas City, Missouri. Union Bank made loans to PC, subject to a perfected security interest in its equipment and other assets, including...
-
The ultimate analysis of a No.4 fuel oil is 86.47 wt% carbons. 11.65% hydrogen, 1.35% sulfur, and the balance noncombustible inerts this oil is burned in a steam-generating furnace with 15% excess...
-
In an enzyme-catalyzed reaction with stoichiometry A B, A is consumed at a rate given by an expression of the MichaelisMenten form: where CA (mol/L) is the reactant concentration, and k1 and k2...
-
If carbon dioxide is cooled at I atm. it condenses directly to a solid (dry ice) at 78.4C. The heat of sublimation at this temperature is H sub (78.4C) = 6030 cal/mol. (a) Calculate the heat removal...
-
A slot machine is a gambling device that the user inserts money into and then pulls a lever (or presses a button). The slot machine then displays a set of random images. If two or more of the images...
-
Design a number guessing game program. The program should generate a random number and then ask the user to guess the number. Each time the user enters his or her guess, the program should indicate...
-
As an illustration of the difficulty in identifying monopolies, try to decide which of the following are monopolies: a train operating company; your local evening newspaper; the village hairdresser;...

Study smarter with the SolutionInn App


