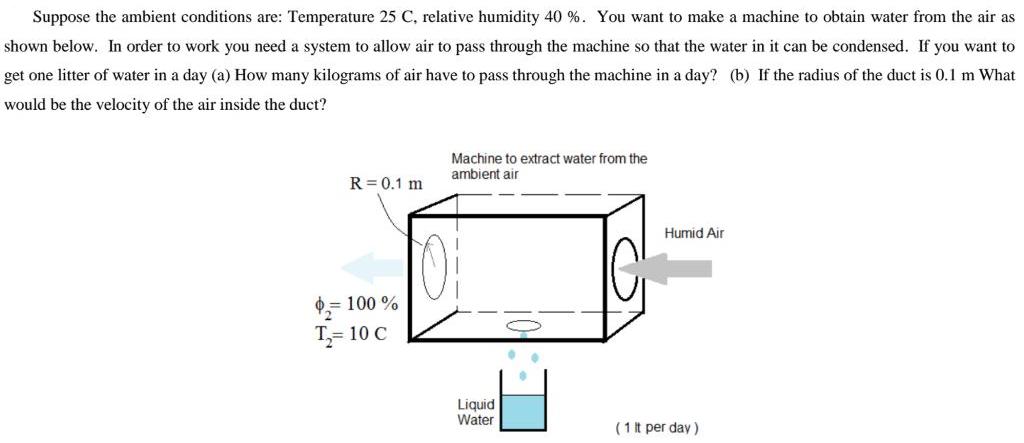
Suppose the ambient conditions are: Temperature 25 C, relative humidity 40 %. You want to make...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
Suppose the ambient conditions are: Temperature 25 C, relative humidity 40 %. You want to make a machine to obtain water from the air as shown below. In order to work you need a system to allow air to pass through the machine so that the water in it can be condensed. If you want to get one litter of water in a day (a) How many kilograms of air have to pass through the machine in a day? (b) If the radius of the duct is 0.1 m What would be the velocity of the air inside the duct? Machine to extract water from the ambient air R= 0.1 m Humid Air = 100 % T,= 10 C Liquid Water (1 t per day) Suppose the ambient conditions are: Temperature 25 C, relative humidity 40 %. You want to make a machine to obtain water from the air as shown below. In order to work you need a system to allow air to pass through the machine so that the water in it can be condensed. If you want to get one litter of water in a day (a) How many kilograms of air have to pass through the machine in a day? (b) If the radius of the duct is 0.1 m What would be the velocity of the air inside the duct? Machine to extract water from the ambient air R= 0.1 m Humid Air = 100 % T,= 10 C Liquid Water (1 t per day)
Expert Answer:

Related Book For 

Principles of heat transfer
ISBN: 978-0495667704
7th Edition
Authors: Frank Kreith, Raj M. Manglik, Mark S. Bohn
Posted Date:
Students also viewed these physics questions
-
A heat exchanger performs as shown below in the Figure A for safety reasons. An engineer suggests that it would be wise to double the heat transfer area so as to double the heat transfer rate. The...
-
You have 1.0kg of copper and want to make a practical solenoid that produces the greatest possible magnetic field for a given voltage should you make your copper wire ling and thin, short and fat, or...
-
You want to make up 3.00 L of aqueous hydrochloric acid, HCl(aq), that has a pH of 2.00. How many grams of concentrated hydrochloric acid will you need? Concentrated hydrochloric acid contains 37.2...
-
A person is pulling on a rope attached to a locomotive of mass m = 100 tons (Fig. 1, right). The person is pulling with a constant force F that is 2 times greater than their body weight, at an upward...
-
A fraction nonconforming control chart has center line 0.01, UCL = 0.0399, LCL = 0, and n = 100. If three-sigma limits are used, find the smallest sample size that would yield a positive lower...
-
If the rate at which energy is dissipated by resistor 1 in Figure P31.86 is \(0.75 \mathrm{~W}\), and \(R_{1}=12 \Omega, \mathscr{E}_{1}=4.5 \mathrm{~V}\), and \(\mathscr{E}_{2}=8.0 \mathrm{~V}\) (a)...
-
Havens Corp. is planning to issue long-term bonds payable to borrow for a major expansion. The chief executive, Richie Havens, asks your advice on some related matters, as follows: a. At what type of...
-
During the current year, merchandise is sold for $3,750,000. The cost of the merchandise sold is $2,550,000. a. What is the amount of the gross profit? b. Compute the gross profit percentage (gross...
-
Barbara was recently widowed, and she subsequently gave her daughter an existing $ 1 0 0 , 0 0 0 life insurance policy under which Barbara was insured, as a gift. Her daughter named herself the...
-
Simtek projects year-end EBIT of $800M which is expected to grow at 5% annually, and capital expenditures will equal depreciation. Simtek has $4,590M of 6% perpetual debt trading at face value and no...
-
Jazel Business services sells its goods in containers which are returnable. These containers are purchased by the company at Sh.20 per container but each container is written down to the book value...
-
Abernathy Corporation was organized on January 1, 2015. It is authorized to issue 10,000 shares of 8%, \($50\) par value preference shares, and 500,000 shares of no-par ordinary shares with a stated...
-
Accounting, Analysis, and Principles On January 1, 2015, Garner issued 10-year, 200,000 face value, 6% bonds at par. Each 1,000 bond is convertible into 30 shares of Garner 2, par value, ordinary...
-
Kennedy Company has the following portfolio of trading investments at December 31, 2015. On December 31, 2016, Kennedys portfolio of trading investments consisted of the following investments. At the...
-
On January 1, 2015, Jennings Company purchased at par 10% bonds having a maturity value of 300,000. They are dated January 1, 2015, and mature January 1, 2020, with interest receivable December 31 of...
-
Assume the same information in E16-1, except that Angela Corporation converts its convertible bonds on January 1, 2016. Instructions (a) Compute the carrying value of the bond payable on January 1,...
-
Three children must make fair division of a painting and sculpture left them by their mother. Using the Knaster Inheritance Procedure, the value each child places on the objects is shown below....
-
During registration at Tech every quarter, students in the Department of Management must have their courses approved by the departmental advisor. It takes the advisor an average of 4 minutes...
-
To decrease the size of personal computer mother boards, designers have turned to a more compact method of mounting memory chips on the board. The single in-line memory modules, as they are called,...
-
Using the information in Problem 1.20, estimate the ambient air temperature that could cause frostbite on a calm day on the ski slopes. From Problem 1.20Suppose that the inner temperature of a 3 mm...
-
A one-tube pass cross-flow heat exchanger is considered for recovering energy from the exhaust gases of a turbine-driven engine. The heat exchanger is constructed of flat plates, forming an egg-crate...
-
A pipe branches symmetrically into two legs of length \(L\), and the whole system rotates with angular speed \(\omega\) around its axis of symmetry. Each branch is inclined at angle \(\alpha\) to the...
-
For the rotating sprinkler of Example 4.13, what value of \(\alpha\) will produce the maximum rotational speed? What angle will provide the maximum area of coverage by the spray? Draw a velocity...
-
Compressed air is stored in a pressure bottle with a volume of \(100 \mathrm{~L}\), at \(500 \mathrm{kPa}\) absolute and \(20^{\circ} \mathrm{C}\). At a certain instant, a valve is opened and mass...

Study smarter with the SolutionInn App


