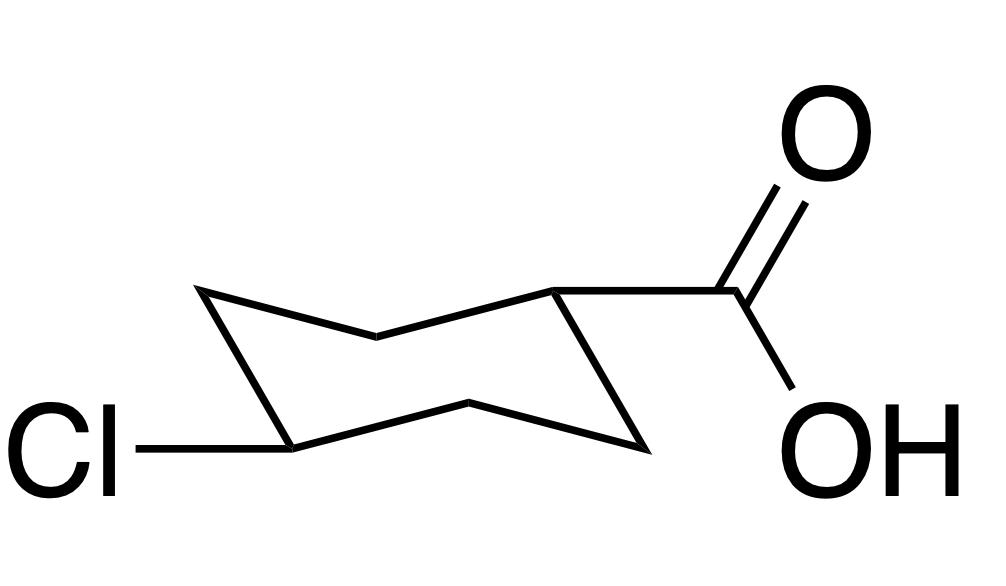
Convert the chair isomer into a flat cyclohexane representation (being sure to preserve the stereochemical information). b)
Fantastic news! We've Found the answer you've been seeking!
Question:
Convert the chair isomer into a flat cyclohexane representation (being sure to preserve the stereochemical information).
b) draw the mechanism for deprotonating the acidic functional group found in your molecule and the final product. (give me an explanation too please)
c) Show the acid/base mechanism for the deprotonation of your acid functional group
D) state what the pKa of the functional group you are deprotonating explain what the arrows were showing using appropriate terminology.
Related Book For 

Business Statistics
ISBN: 978-0321925831
3rd edition
Authors: Norean Sharpe, Richard Veaux, Paul Velleman
Posted Date:




