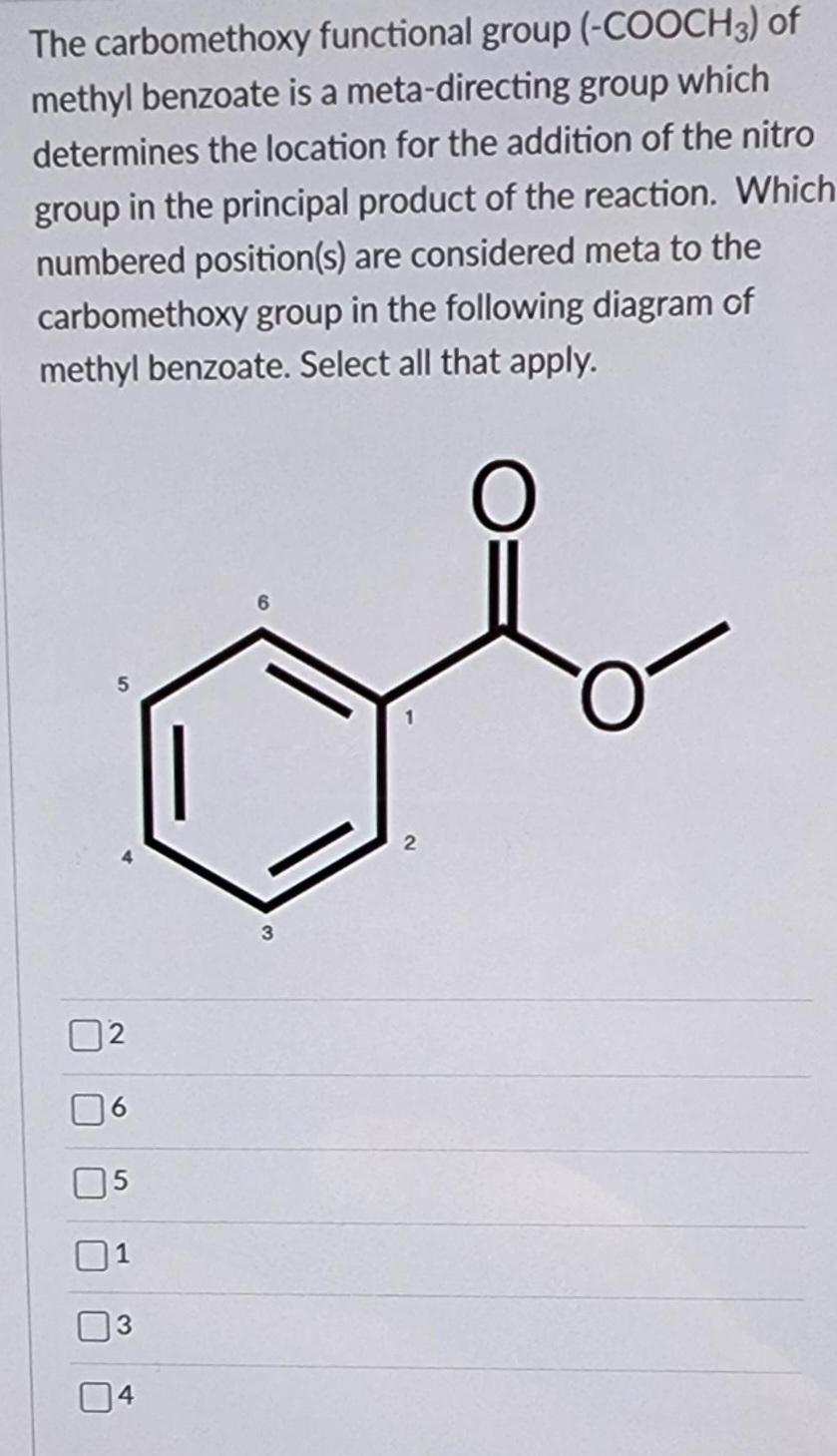
The carbomethoxy functional group (-COOCH3) of methyl benzoate is a meta-directing group which determines the location...
Fantastic news! We've Found the answer you've been seeking!
Question:
Transcribed Image Text:
The carbomethoxy functional group (-COOCH3) of methyl benzoate is a meta-directing group which determines the location for the addition of the nitro group in the principal product of the reaction. Which numbered position(s) are considered meta to the carbomethoxy group in the following diagram of methyl benzoate. Select all that apply. 3 2 The carbomethoxy functional group (-COOCH3) of methyl benzoate is a meta-directing group which determines the location for the addition of the nitro group in the principal product of the reaction. Which numbered position(s) are considered meta to the carbomethoxy group in the following diagram of methyl benzoate. Select all that apply. 3 2
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Select all that apply to the Diamond Problem: C++ Compilers are equipped to handle this automatically with no further work from the programmer It can only occur when in the inheritance chain of an...
-
Apply Hammond's postulate to decide which reaction is faster: addition of HBr to 2 methylpropene or addition of HBr to trans-2-butene. Assume that the energy difference between the starting alkenes...
-
Which can lose a proton more readily, a methyl group bonded to cyclohexane or a methyl group bonded to benzene? (11 CH3
-
Describe two methods for determining the molecular mass of a polypeptide. Which is more accurate and why?
-
1. Pricing below production cost or selling at prices in foreign markets less than those in domestic markets is known as __________. 2. Under global trade rules, the United States was allowed to ban...
-
Refer to the information provided in M4?13. Barry Gold, a tax client of Sunrise Accounting, requires 20 miles of transportation, 50 hours of processing time, and 3 hours of office support. Using the...
-
Describe which characteristics of HR metrics and workforce analytics are likely to result in greater return on investment and organizational impact.
-
A long cylindrical rod of very high emissivity is heat treated within a long, evacuated oven of square cross section as shown in the sketch. The oven walls exhibit blackbody behavior. Due to...
-
If there were a shift of $8,000 in sales revenue from the banquet area to the dining room, would you expect the restaurant's overall operating income to increase or decrease? Explain your reasoning...
-
Fitz Products Inc. reported $1,075,049 profit in 2023 and declared preferred dividends of $75,100. The following changes in common shares outstanding occurred during the year. Jan. 1 78,000 common...
-
Match each type of reaction to a proposed change in the organization with its corresponding description. Those who engage in this reaction may openly object to the change and even sabotage the...
-
In Problems 4-13, decide whether the statement is true or false. If it is false, tell what is wrong. In *Row and *Row + notation, the target row is the last number listed.
-
A plane travels \(200 \mathrm{mph}\) relative to the ground while flying with a strong wind and only \(150 \mathrm{mph}\) returning against it. What is the plane's speed in still air?
-
In Problems 9-16, find the indicated matrices, if possible. \[[\mathrm{A}]=\left[\begin{array}{rr}1 & 2 \\4 & 0 \\-1 & 3 \\2 & 1\end{array}ight] \quad[\mathrm{B}]=\left[\begin{array}{rr}4 & 2 \\-1 &...
-
The demand for a product varies from 150,000 units at \(\$ 110\) per unit to 300,000 at \(\$ 20\) per unit. Also, 300,000 could be supplied at \(\$ 90\) per unit, whereas only 200,000 could be...
-
Describe the addition method for solving a system of equations.
-
Consider the following option strategy where the options are all for the same stock that does not pay a dividend and all the options have the same maturity: Long one call with $100 strike price...
-
Pearson Education, a publisher of college textbooks, would like to know if students prefer traditional textbooks or digital textbooks. A random sample of students was asked their preference and the...
-
When ethene is mixed with hydrogen in the presence of a platinum catalyst, hydrogen adds across the double bond to form ethane. At room temperature, the reaction goes to completion. Predict the signs...
-
The pKa of ascorbic acid (vitamin C, page 2) is 4.17, showing that it is slightly more acidic than acetic acid (CH3COOH, pKa, 4.74). (a) Show the four different conjugate bases that would be formed...
-
Propose a Williamson synthesis of 3-butoxy-1, 1-dimethylcyclohexane from 3, 3-dimethylcyclohexanol and butan-1-ol.
-
Determine the probability mass function of \(X\) from the following cumulative distribution function: \[ F(x)=\left\{\begin{array}{lr} 0 & x. \] Figure 3.3 displays a plot of \(F(x)\). From the plot,...
-
Two new product designs are to be compared on the basis of revenue potential. Marketing believes that the revenue from design A can be predicted quite accurately to be \(\$ 3\) million. The revenue...
-
The probability that a wafer contains a large particle of contamination is 0.01. If it is assumed that the wafers are independent, what is the probability that exactly 125 wafers need to be analyzed...

Study smarter with the SolutionInn App



