1- Explain why all the amino acids except for glycine are chiral? 2-Identify the amino acids...
Fantastic news! We've Found the answer you've been seeking!
Question:
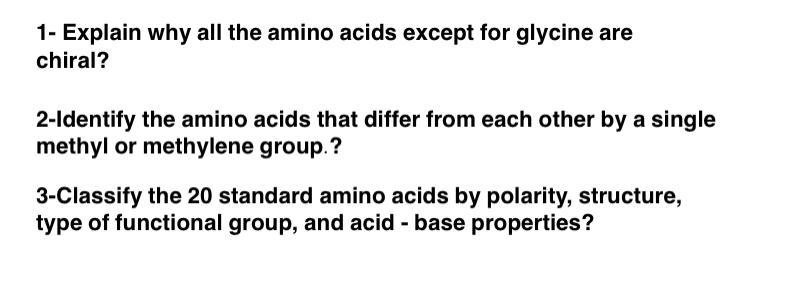
Transcribed Image Text:
1- Explain why all the amino acids except for glycine are chiral? 2-Identify the amino acids that differ from each other by a single methyl or methylene group.? 3-Classify the 20 standard amino acids by polarity, structure, type of functional group, and acid - base properties? 1- Explain why all the amino acids except for glycine are chiral? 2-Identify the amino acids that differ from each other by a single methyl or methylene group.? 3-Classify the 20 standard amino acids by polarity, structure, type of functional group, and acid - base properties?
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemical engineering questions
-
Global warming and climate change forecasts call for an increase in sea levels which require a Climate Adaptation and Mitigation plan for the Airport's bayside properties. SFO has previously secured...
-
A major component of olive oil is glyceryl trioleate. (a) Give the structures of the products expected from the saponification of glyceryl trioleate with excess aqueous NaOH. (b) Give the structure...
-
A major component of financial planning is to forecast future financial statements. If you had a company's balance sheets and income statements for the past 5 years but no other information, how...
-
Why do we use the absolute value of x or of g(x) in the derivative formulas for the natural logarithm?
-
A process called helium burning is thought to occur inside older stars, forming carbon: 34He ( 12C If the reaction proceeds with 0.00781 g of mass lost on a molar basis, how much energy is given off?
-
In terms of leadership behaviors, someone who focuses on doing a very good job of planning work tasks, setting performance standards, and monitoring results would be described as _________. (a) task...
-
Reliable Professionals Pty Ltd is a firm that offers a wide variety of professional services such as marketing, employment of professionals for other firms and accounting services. The firm has the...
-
Your supervisor has asked you to research the following situation concerning Shanita Mauer. Shanita worked and lived in Los Angeles, CA. She accepted a new position with her firm in Dallas, TX....
-
Laura mentions that employees will come back from tea break on time because they know it will let down their fellow work mates if they don't. Imagine an employee named David frequently does not...
-
A beam ABCD consisting of a simple span BD and an overhang AB is loaded by a force P acting at the end of the bracket CEF (see figure). (a) Determine the deflection δA at the end of the...
-
July. 15 Declared a cash dividend payable to common stockholders of $162,000. Aug. 15 Date of the record is August 15 for the cash dividend declared on July 15. Aug. 31 Paid the dividend declared on...
-
Find and describe a Web site for a health club. How does the information relate to the consumer decision- making process to join the club?
-
How do rational choice theorists explain addictive behaviour as a rational choice, and are their hypotheses explained by the econometric evidence? What are some of the problems with this econometric...
-
Find and describe two sports Web sites that specifically appeal to children. How does this information relate to the process of consumer socialization?
-
What are nifs and sophisciates and how do the ways they deal with present bias differ? Illustrate with some examples.
-
Are people always an organizations most valuable asset? Why or why not? Suppose your boss asked you to summarize the major peoplerelated concerns related to opening an office in India. What issues...
-
A 10 g bullet is fired into a 1 kg block of wood, where it lodges. The block then slides 4 m across a wood floor before coming to a stop (use k = 0.2 for wood on wood). A) What was the bullet's...
-
X-1 Find the domain of the function f(x) : x 1 2 - O (-00, -1) U (-1, ) O (-00, 1) U (1, ) O -00, -1) U (-1, 1) U (1, 0) O (- 1, 1)
-
What is dn for each of the following complexes? Pd(PPh3)4
-
The ==C-H stretching absorption of 2-methyl- I-pentene is observed at 3090 cm-1. If the hydrogen were replaced by deuterium, at what wavenumber would the ==C-D stretching absorption be observed?...
-
(a) Give a curved-arrow mechanism for the reaction in Eq. 10.6. (b) After reading Study Guide Link 10.2, explain why the rearrangement in Eq. 10.7 is favorable even though both of the carbocation...
-
Determine the value of the shape-efficiency factor \(\phi_{B}^{f}\) for strength-limited design in bending using the dimensions shown on the diagrams a. For the tube-section shown in Fig. E10.5(A) b....
-
A beam of length \(L\), loaded in bending, must support a specified bending moment \(M\) without failing and be as light as possible. Show that to minimize the mass of the beam per unit length, \(m /...
-
The elastic shape factor measures the gain in stiffness by shaping, relative to a solid square section of the same cross-section area and thus mass per unit length. Shape factors can be determined by...

Study smarter with the SolutionInn App



