General Chemistry 4th Edition McQuarrie Rock Gallogly Initial rate data at a certain temperature is given...
Fantastic news! We've Found the answer you've been seeking!
Question:
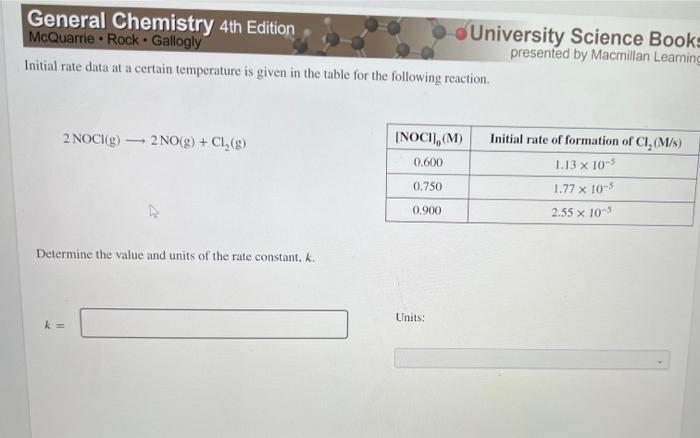
Transcribed Image Text:
General Chemistry 4th Edition McQuarrie Rock Gallogly Initial rate data at a certain temperature is given in the table for the following reaction. 2 NOCI(g) 2NO(g) + Cl₂(g) - Determine the value and units of the rate constant. k. k= [NOCI], (M) 0.600 0.750 0.900 Units: University Science Books presented by Macmillan Learning Initial rate of formation of Cl, (M/s) 1.13 x 10-5 1.77 x 10-5 2.55 x 10-5 General Chemistry 4th Edition McQuarrie Rock Gallogly Initial rate data at a certain temperature is given in the table for the following reaction. 2 NOCI(g) 2NO(g) + Cl₂(g) - Determine the value and units of the rate constant. k. k= [NOCI], (M) 0.600 0.750 0.900 Units: University Science Books presented by Macmillan Learning Initial rate of formation of Cl, (M/s) 1.13 x 10-5 1.77 x 10-5 2.55 x 10-5
Expert Answer:
Answer rating: 100% (QA)
Rafe equation Rafe where Now using 1 eq 11 the table p... View the full answer

Related Book For 

Financial Algebra advanced algebra with financial applications
ISBN: 978-0538449670
1st edition
Authors: Robert K. Gerver
Posted Date:
Students also viewed these chemistry questions
-
New Horizons Airlines wants to forecast the mean number of unoccupied seats per flight to Germany next year. To develop this forecast, the records of 49 flights are randomly selected from the files...
-
The Consumer Credit Counseling Service suggests that the monthly food budget be between 15-30% of income. a. What is Laura's total monthly food bill including dining out? b. What percent of her...
-
What is the Consumer Credit Counseling Services (CCCS)?
-
Refer to the Webers Data Set above. 1. Compute the direct materials efficiency variance for onion, mustard, and ketchup. 2. As a manager, what would you learn from the variances and supporting data?...
-
Steve is a member of a local church. May he deduct as a charitable contribution the commuting expenses for the Sundays that he is assigned to usher?
-
John and Carlos are utilitarians. John believes that labour supply is highly elastic, whereas Carlos believes that labour supply is quite inelastic. How do you suppose their views about income...
-
During its first year of operations, Atlas Travel earned revenue of $400,000 on account. Industry experience suggests that Atlas Travels uncollectible accounts will amount to 2% of revenues. On...
-
The weekly demand (in cases) for a particular brand of automatic dishwasher detergent for a chain of grocery stores located in Columbus, Ohio, follows. a. Construct a time series plot. What type of...
-
Companies that have preferred stock outstanding promise to pay a stated dividend for an infinite period. Preferred stock is treated like a perpetuity if the payments last forever. Preferred stocks...
-
John Fuji (birthdate June 6, 1981) moved from California to Washington in December 2018. His earnings and income tax withholding for 2019 for his job as a manager at a Washington apple-processing...
-
In service partnerships, if properly booked for tax purposes pursuant to Treasury Regulations under IRC 731, partners who receive so-called "draw" allowances are not deemed to receive distributions...
-
Mention the different criteria for phase equilibrium. What is the necessity for a system to be in equilibrium?
-
What do you mean by criterion for equilibrium of a system?
-
How can equilibrium be classified? When does a system reach equilibrium?
-
Explain the phase rule for non-reacting system. State Duhem's theorem in substantiating the phase rule.
-
Justify the statement: Fugacities of a pure component in vapour and liquid phases are identical.
-
1. In a deposit of sand ('=35), the unit weight above the water table (dry) is yd=17 kN/m, unit weight below the water table (saturated) is ysat=20.51 kN/m. if the water table is at depth of 2.5 m...
-
Suppose the concentration of glucose inside a cell is 0.1 mm and the cell is suspended in a glucose solution of 0.01 mm. a. What would be the free energy change involved in transporting 10-o mole of...
-
In 2008, the maximum taxable income for Social Security was $102,000 and the tax rate was 6.2%. a. What is the maximum Social Security tax anyone could have paid in the year 2008? b. Randy had two...
-
Use the tax computation worksheet for a head of household taxpayer's taxable income and y represent the tax. Express each line of the worksheet as a linear equation in y = mx + b form. Use interval...
-
Graph the profit function. Determine the coordinates of the maxi- mum point of the profit graph. At what price, p, is profit maximized? Round to the nearest cent. This will be the price at which...
-
The following report was prepared by M. Salvemini, CPA, to accompany unaudited financial statements: We have prepared the statement of net worth, dated November 30, 19X1, that is attached hereto....
-
W.E. Jolson, CPA, has issued the following report on the Bambino Lumber Company's balance sheet: I have audited the balance sheet of Bambino Lumber Co. for the period ended December 31, 19X1. Due to...
-
You have been asked to prepare unaudited financial statements for the Marek Company for the year ended December 31, 19X5. Marek does not want the notes to financial statements to disclose that the...

Study smarter with the SolutionInn App


