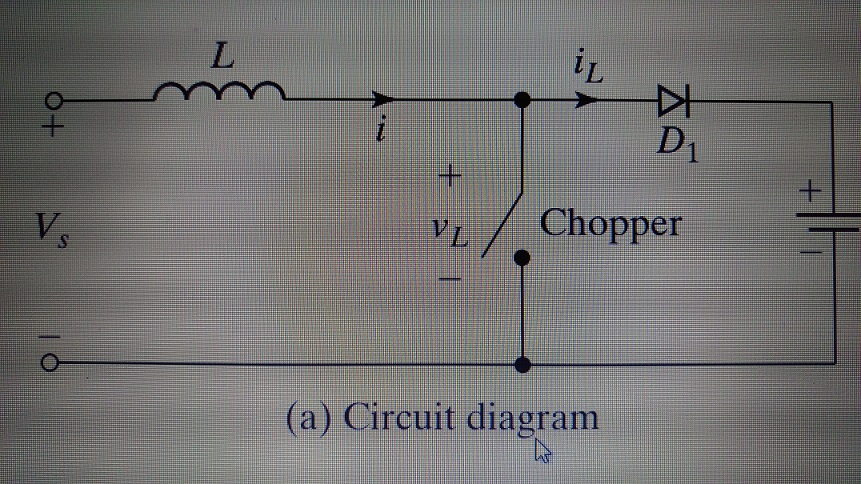
V 5.4 The dc converter shown in Figure 5.8a is used to control power flow from...
Fantastic news! We've Found the answer you've been seeking!
Question:

Transcribed Image Text:
V₂ 5.4 The dc converter shown in Figure 5.8a is used to control power flow from a dc voltage, 110V to a battery voltage, E = 220 V. The power transferred to the battery is 25 kW. The current ripple of the inductor is negligible. Determine (a) the duty cycle K, (b) the effective load resistance Req, and (c) the average input current Is. = 6+ Vs IO L MI + VL iL # D₁ Chopper (a) Circuit diagram + V₂ 5.4 The dc converter shown in Figure 5.8a is used to control power flow from a dc voltage, 110V to a battery voltage, E = 220 V. The power transferred to the battery is 25 kW. The current ripple of the inductor is negligible. Determine (a) the duty cycle K, (b) the effective load resistance Req, and (c) the average input current Is. = 6+ Vs IO L MI + VL iL # D₁ Chopper (a) Circuit diagram +
Expert Answer:

Related Book For 

Posted Date:
Students also viewed these computer network questions
-
A combinational circuit is used to control a seven-segment display of decimal digits, as shown in Figure 20.36. The circuit has four inputs, which provide the four-bit code used in packed decimal...
-
A two bin approach is used to control inventory for a certain low cost hardware item. Each bin holds 500 units of the item. When one bin becomes empty, an order for 500 units is released to replace...
-
A closed-loop negative unity feedback system is used to control the yaw of the A-6 Intruder attack jet. When the loop transfer function is determine (a) the root locus breakaway point and (b) the...
-
2. Two point charges are located at two comers of a triangle as shown. What is the electric potential at the right corner of the triangle? A. 21 10 V C. 4.5 x 10 V B. 3.4 x 10 V D. 6.3 x 10 V 10 em...
-
Use the frequency distribution for Exercise 23 to make a relative frequency distribution. Round to the nearest tenth percent. In exercise 32 23. Use the test scores of 32 students taking Business 205...
-
Kostrivas Company has gathered the following information. Units in beginning work in process ........ _0_ Units started into production .......... 40,000 Units in ending work in process ...........
-
Nationwide Insurance developed a program to recruit new insurance agents by offering planning, training, and start-up financing to build self-sustaining agencies. These new agents would be...
-
Debt Service Fund Trial Balance. Following is Franklin Countys debt service fund pre-closing trial balance for the fiscal year ended June 30, 2011. Required Using information provided by the trial...
-
(5) Strong Data Processing Inequality for the Divergence. Given finite alphabets X and Y, consider a conditional probability distribution W(y|x), for (x, y) Xxy, such that for some y. y, W (y, x) > c...
-
The following table summarizes the operating results for Bene Petits first year of operations: Bene Petit First year operating data: Single (1 serving) Dual (2 servings) Family (4 servings) Total...
-
Write an essay on A further understanding of Mindsets
-
Discuss the limitations of fiscal policy during a recession. What fiscal policy has been used during previous recessionary periods? How does the fiscal policy during the COVID-19 recession differ...
-
Does culture affect how you can respond to a difficult negotiation?
-
What dates/deadline can the Company specify in the Invitation? __________________________________________________________________________________...
-
One of the key factors impacting International Human Resource Management is technology. Thetechnological space has mainly been driven by emerging technologies such as artificial intelligence. Given...
-
The Shoot for the Cure: 3-on-3 Basketball Tournament, organized by the American Cancer Society (ACS) and coordinated by Mark, the Event Manager, was going smoothly. The games were running on schedule...
-
(a) If the potential energy of electron in the state 32 of hydrogen atom is -1.7eV, calculate its kinetic 1 energy. Hint: E =-13.6eV- (b) What are the quantum numbers for the first state and second...
-
What mass of H2 will be produced when 122 g of Zn are reacted? Zn(s) + 2HCl(aq) ( ZnCl2(aq) + H2(g)
-
An RL circuit is shown in Figure PI 1.21. (a) Select the two stable variables and obtain the vector differential equation where the output is v0 (t). (b) Determine whether the state variables are...
-
In Figure E4.12, consider the closed-loop system with measurement noise N(s), where In the following analysis, the tracking error is defined to be E(s) = R(s) - Y(s): (a) Compute the transfer...
-
Consider the closed-loop system in Figure AP6.5. Suppose that all gains are positive, that is, K1 > 0, K2 > 0, K3 > 0, K4 > 0, and K5 > 0. (a) Determine the closed-loop transfer function T(s) =...
-
In the section of his 2007 letter to the shareholders of Berkshire Hathaway titled Fanciful FiguresHow Public Companies Juice Earnings, Warren Buffett referred to the investment return assumption...
-
Based on 2012 revenues, the six largest providers of oilfield services are: 1. Schlumberger Ltd. (NYSE: SLB) Revenues: $42.1 billion Net income: $5.5 billion 2. Halliburton (NYSE: HAL) Revenues:...
-
On 21 September 2000, Intel Corporation (NASDAQ -GS: INTC)3 issued a press release containing information about its expected revenue growth for the third quarter of 2000. The announced growth fell...

Study smarter with the SolutionInn App


