The Decomposition of Nitrogen Dioxide Introduction The applications of chemistry focus primarily on chemical reactions, processes...
Fantastic news! We've Found the answer you've been seeking!
Question:

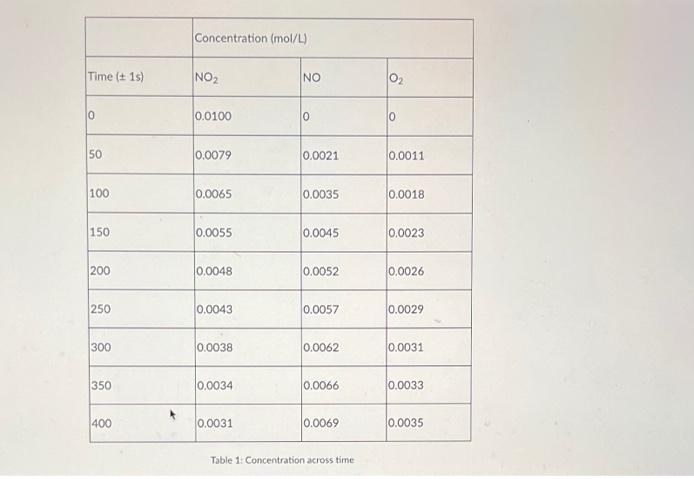

Transcribed Image Text:
The Decomposition of Nitrogen Dioxide Introduction The applications of chemistry focus primarily on chemical reactions, processes that are defined by their reactants (substances that undergo change in a reaction) and products (substances that are formed as a result of a reaction). In order to be useful, reactions must occur at a reasonable rate. It is important, then, to understand the factors that govern the rate of a reaction. This is where chemical kinetics comes into play: it is the area of chemistry that concerns reaction rates. Perhaps unsurprisingly, the study of reaction rates depends heavily on calculus. In this project, we will apply our knowledge of rates of change to the decomposition of reactants in chemical reactions to gain knowledge of the chemical kinetics at work. Note that for this project, no knowledge of chemistry is required. One of the goals is to see how we can use the math we are learning in this course to answer questions that may arise in other fields. As you work on this project, keep the following guiding question in mind: Focus Question: In chemistry, they have the notion of an instantaneous reaction rate of a reactant/product. What is this idea called in mathematics? Project Description Nitrogen dioxide, a gas that causes air pollution, decomposes into nitric oxide and oxygen according to the following equation: 2NO₂(g) → 2NO(g) + O₂(g). Suppose in an experiment we start with a flask of nitrogen dioxide at room temperature and quickly heat it up to 300°C, where it decomposes according to the above equation. We then measure the concentration of nitrogen dioxide, nitric oxide, and oxygen over time as the nitrogen dioxide decomposes. The results of this experiment are summarized in the table below. Using this data, answer the questions below. Time (± 1s) 0 50 100 150 200 250 300 350 400 Concentration (mol/L) NO₂ 0.0100 0.0079 0.0065 0.0055 0.0048 0.0043 0.0038 0.0034 0.0031 NO 0 0.0021 0.0035 0.0045 0.0052 0.0057 0.0062 0.0066 0.0069 Table 1: Concentration across time 0₂ 0 0.0011 0.0018 0.0023 0.0026 0.0029 0.0031 0.0033 0.0035 1. Using the data above given on Table 1, create a data plot with time on the x-axis and concentration of chemicals on the y-axis. You will have three graphs on the same plot, one for each of the involved chemicals. Be sure to label and title your graph appropriately. Check the guides on how to create a plot using Excel or Google Sheets. 2. Draw a line connecting the points (0, 0.0100) and (400, 0.0031) on the graph of NO₂ concentration over time. What is the slope of this line? What are the units? What does it represent? Note: In the language of chemistry, this question is asking you to compute the average rate at which the concentration of NO₂ changes during the first 400 seconds of the reaction. 3. Given that chemical reaction rates tend to slow down over time, do you expect the instantaneous change in concentration of NO₂ when t-0 to be greater than, less than, or equal to the slope computed in number 2? Similarly, do you expect the instantaneous change of concentration of NO₂ at t-400 to be greater than, less than, or equal to the slope computed in number 2? Explain your reasoning. 4. Use the data and your plot to estimate the instantaneous rate of change of concentration of NO₂, NO, and O₂ when t=100. What are the units for these numbers that are calculated? What do these values represent in the context of this problem? Note: In the language of chemistry, this question is asking you to estimate the instantaneous reaction rate of NO₂, NO, and O₂ 100 seconds into the reaction. The Decomposition of Nitrogen Dioxide Introduction The applications of chemistry focus primarily on chemical reactions, processes that are defined by their reactants (substances that undergo change in a reaction) and products (substances that are formed as a result of a reaction). In order to be useful, reactions must occur at a reasonable rate. It is important, then, to understand the factors that govern the rate of a reaction. This is where chemical kinetics comes into play: it is the area of chemistry that concerns reaction rates. Perhaps unsurprisingly, the study of reaction rates depends heavily on calculus. In this project, we will apply our knowledge of rates of change to the decomposition of reactants in chemical reactions to gain knowledge of the chemical kinetics at work. Note that for this project, no knowledge of chemistry is required. One of the goals is to see how we can use the math we are learning in this course to answer questions that may arise in other fields. As you work on this project, keep the following guiding question in mind: Focus Question: In chemistry, they have the notion of an instantaneous reaction rate of a reactant/product. What is this idea called in mathematics? Project Description Nitrogen dioxide, a gas that causes air pollution, decomposes into nitric oxide and oxygen according to the following equation: 2NO₂(g) → 2NO(g) + O₂(g). Suppose in an experiment we start with a flask of nitrogen dioxide at room temperature and quickly heat it up to 300°C, where it decomposes according to the above equation. We then measure the concentration of nitrogen dioxide, nitric oxide, and oxygen over time as the nitrogen dioxide decomposes. The results of this experiment are summarized in the table below. Using this data, answer the questions below. Time (± 1s) 0 50 100 150 200 250 300 350 400 Concentration (mol/L) NO₂ 0.0100 0.0079 0.0065 0.0055 0.0048 0.0043 0.0038 0.0034 0.0031 NO 0 0.0021 0.0035 0.0045 0.0052 0.0057 0.0062 0.0066 0.0069 Table 1: Concentration across time 0₂ 0 0.0011 0.0018 0.0023 0.0026 0.0029 0.0031 0.0033 0.0035 1. Using the data above given on Table 1, create a data plot with time on the x-axis and concentration of chemicals on the y-axis. You will have three graphs on the same plot, one for each of the involved chemicals. Be sure to label and title your graph appropriately. Check the guides on how to create a plot using Excel or Google Sheets. 2. Draw a line connecting the points (0, 0.0100) and (400, 0.0031) on the graph of NO₂ concentration over time. What is the slope of this line? What are the units? What does it represent? Note: In the language of chemistry, this question is asking you to compute the average rate at which the concentration of NO₂ changes during the first 400 seconds of the reaction. 3. Given that chemical reaction rates tend to slow down over time, do you expect the instantaneous change in concentration of NO₂ when t-0 to be greater than, less than, or equal to the slope computed in number 2? Similarly, do you expect the instantaneous change of concentration of NO₂ at t-400 to be greater than, less than, or equal to the slope computed in number 2? Explain your reasoning. 4. Use the data and your plot to estimate the instantaneous rate of change of concentration of NO₂, NO, and O₂ when t=100. What are the units for these numbers that are calculated? What do these values represent in the context of this problem? Note: In the language of chemistry, this question is asking you to estimate the instantaneous reaction rate of NO₂, NO, and O₂ 100 seconds into the reaction.
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The decomposition of nitrogen dioxide (NO2) into nitrogen monoxide (NO) and oxygen is a second-order reaction. This means that the concentration C of NO2 at time t is given by 1/C = kt + 1/C0, where...
-
The decomposition of nitrogen dioxide, 2NO2(g) 2NO(g)+ O2(g) has a rate constant of 0.498 M/s at 319oC and a rate constant of 1.81 M/s at 354oC. What are the values of the activation energy and the...
-
In this project we will apply what we learned about capital budgeting using real-world examples. General Instructions: This is an individual project, i.e., each one of you will have to submit the...
-
Regulation imperial soccer fields have a length of 91 meters and a width of 39 meters. Show that during a friendly match there are always two players that are not farther from each other than 19...
-
The following is a 2-way ANOVA conducted using our final project data base. Sex and Treatment Condition are your independent variables, and Post-Treatment Anxiety (RCMAS) is your dependent variable....
-
Each individual in a sample was asked to indicate on a quantitative scale how willing he or she was to spend money on the environment and also how strongly he or she believed in God (Religion and...
-
Prepare a balance sheet for Anzac Properties, based in Australia, from the following information. What is the net working capital and debt ratio? Cash A$50,000 Accounts Receivable 43,800 Accounts...
-
Ehler Corporation sells rock-climbing products and also operates an indoor climbing facility for climbing enthusiasts. During the last pan of 2017, Ehler had the following transactions related to...
-
A ( n ) _ _ _ _ _ _ _ _ _ _ contains information about a given person, product, or event. Attribute Column Field Record
-
4. Distinct Items There is a list of items in the shopping cart, each having a cost associated with it. There are n items, the cost of the ith item is / dollars and m items have already been bought...
-
10. What will be the value of i after the C statements below have been executed? i=3; j= 10; if ((3* i)
-
How can focus groups and interviews be used to assess website effectiveness?
-
Describe the different types of formats an online retailer might follow.
-
Briefly evaluate the strengths and weaknesses of affiliate marketing for a well-known retailer.
-
What is meant by distributing services? How can an experience or something intangible be distributed?
-
Why should content development be distributed through a large organisation?
-
Figure below shows the cross section of an embankment to be constructed. For the embankment, y= 110kpa. The soil for the embankment has to be brought from a borrow pit. The soil at the borrow pit has...
-
The pendulum consists of two rods: AB is pin supported at A and swings only in the y-z plane, whereas a bearing at B allows the attached rod BD to spin about rod AB. At a given instant, the rods have...
-
A tire company claims that the lifetimes of its tires average 50,000 miles. The standard deviation of tire lifetimes is known to be 5000 miles. You sample 100 tires and will test the hypothesis that...
-
In a sample of 100 steel wires the average breaking strength is 50 kN, with a standard deviation of 2 kN. a. Find a 95% confidence interval for the mean breaking strength of this type of wire. b....
-
To study the effect of curing temperature on shear strength of a certain rubber compound, 80 specimens were cured at 150C and 95 were cured at 130C. The specimens cured at 150C had an average shear...
-
At what annual rate would the following have to be invested? a. \($820\) to grow to \($1,988.12\) in 13 years b. \($320\) to grow to \($423.10\) in 6 years c. \($57\) to grow to \($290.30\) in 18...
-
To what amount will the following investments accumulate? a. $6,000 invested for 12 years at 12 percent compounded annually b. $7,500 invested for 8 years at 8 percent compounded annually c. $6,400...
-
If youd like to have $5,000,000 at retirement in 45 years and you expect to earn 10 percent annually, which is around the average return over the past 50 years, what lump sum would you have to invest...

Study smarter with the SolutionInn App



