The density of schottky defect in a certain sample of NaCl is 5x10^11 m^11 at 25C....
Fantastic news! We've Found the answer you've been seeking!
Question:
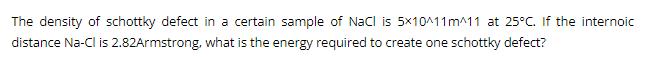
Transcribed Image Text:
The density of schottky defect in a certain sample of NaCl is 5x10^11 m^11 at 25°C. If the internoic distance Na-Cl is 2.82Armstrong, what is the energy required to create one schottky defect? The density of schottky defect in a certain sample of NaCl is 5x10^11 m^11 at 25°C. If the internoic distance Na-Cl is 2.82Armstrong, what is the energy required to create one schottky defect?
Expert Answer:
Answer rating: 100% (QA)
To calculate the energy required to create one Schottky defect in NaCl we can use the formula for the formation energy of a Schottky defect E 2 H G wh... View the full answer

Related Book For 

Essentials of Materials Science and Engineering
ISBN: 978-1111576851
3rd edition
Authors: Donald R. Askeland, Wendelin J. Wright
Posted Date:
Students also viewed these physics questions
-
The fraction of lattice points occupied by vacancies in solid aluminum at 660 C is 10-3. What is the energy required to create vacancies in aluminum?
-
Exercise 5.5 explored the enthalpy representation of thermodynamics for an equilibrium ensemble of systems in contact with a volume bath. Here we extend that analysis to an ensemble out of...
-
The following additional information is available for the Dr. Ivan and Irene Incisor family from Chapters 1-5. Ivan's grandfather died and left a portfolio of municipal bonds. In 2012, they pay Ivan...
-
Let y vary directly with x. Complete the following. Find y when x = 1.3, if y = 7.2 when x = 5.2.
-
On January 1, 1998, the market interest rate is 15%. Friedman Software, Inc. issues $300,000 of 18%, 5-year bonds payable. The bonds pay interest on April 30, August 31, and December 31 (3 times a...
-
Bronson Paper Products purchased 10,000 acres of forested timberland in March 2025. The company paid $1,700 per acre for this land, which was above the $800 per acre most farmers were paying for...
-
How do systems serve the different management groups in a business? Describe the characteristics of transaction processing systems (TPS) and the roles they play in a business. Describe the...
-
Union Express has 60 tons of cargo that needs to be shipped from Boston to Dallas. The shipping capacity on each of the routes Union Express planes fly each night is shown in the following table:...
-
A firm evaluates all of its projects by using the NPV decision rule. At a required return of 14 percent, the NPV for the following independent project is_____and the firm should____the project. Any...
-
Determine which of the following are not well-formed formulas. 1. (S T) (U W) 2. (K L) ( G H) 3. (E F) (W X) 4. (B T) (C U ) 5. (F Q) (A E T) 6. D [(P Q) (T R)] 7. [(D Q) (P ...
-
Burham Industry needs RM 825,471.70 as a minimum cash requirement to support the firm's day-to-day operations. Hence, Burham Industry implement several strategies that could reduce the cash cycle and...
-
Explain the nature of the statement of cash flows and discuss how it can be helpful in identifying cash flow problems.
-
Res ipsa loquitur (the thing speaks for itself or the circumstances speak for themselves) is the legal doctrine that shifts the burden of proof in a negligence case from the plaintiff to the...
-
Discuss the crucial importance of cash to a business.
-
Interpret a statement of cash flows.
-
There are multiple types of evidence that can be presented at trial. Direct evidence Demonstrative evidence Documentary evidence Hearsay evidence
-
What does the process of evaluation in Bloom's Taxonomy primarily involve? Comparing and contrasting information , Not Selected Applying previously learned materials into new situations , Not...
-
Which internal control principle is especially diffi cult for small organizations to implement? Why?
-
The crystal shown in Figure 4-18 contains two dislocations A and B. if a shear stress is applied to the crystal as shown, what will happen to dislocations A and B?
-
Determine the specific yield strengths of the strongest Al, Mg, Cu, Ti, and Ni alloys. Use the densities of the pure metals, in lb/in.3, in your calculations. Try to explain their order.
-
Calculate the density of a honeycomb structure composed of the following elements: The two 2-mm-thick cover sheets are produced using an epoxy matrix prepreg containing 55 vol% E-glass fibers. The...
-
Pier10 Inc. entered into a 5-year lease and recorded a right-of-use asset and lease liability of \(\$ 88,000\) on January 1, 2020. Pier10 Inc. was aware of the lessor's implicit rate of interest of...
-
Lessee Company enters into a 6-year finance lease of non-specialized equipment with Lessor Company on January 1,2020 . Lessee has agreed to pay \(\$ 28,000\) annually beginning immediately on January...
-
Frontier Inc. enters into an 8 -year lease contract to lease equipment with a useful life of 8 years. Annual lease payments are due with the first payment made immediately on January 1, 2020, the...

Study smarter with the SolutionInn App


