Question: The elements in Group 2A are known by what name? alkali metals O noble gases O transition metals alkaline earth metals O halogens How

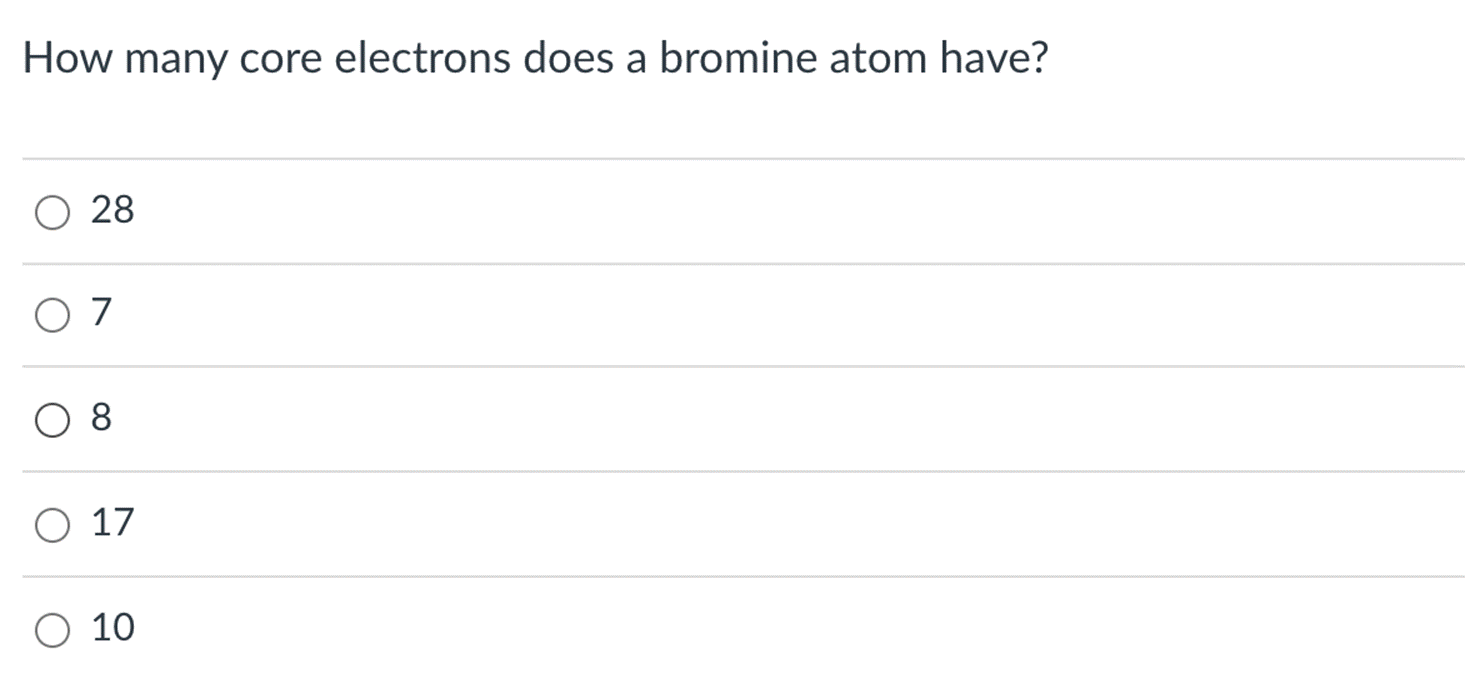
The elements in Group 2A are known by what name? alkali metals O noble gases O transition metals alkaline earth metals O halogens How many core electrons does a bromine atom have? O 28 O 7 O 8 O 17 O 10
Step by Step Solution
3.42 Rating (146 Votes )
There are 3 Steps involved in it
The elements in Grouf 2A are knocon by alkaline eax th metols alkalne Soluions Cbyd... View full answer

Get step-by-step solutions from verified subject matter experts


