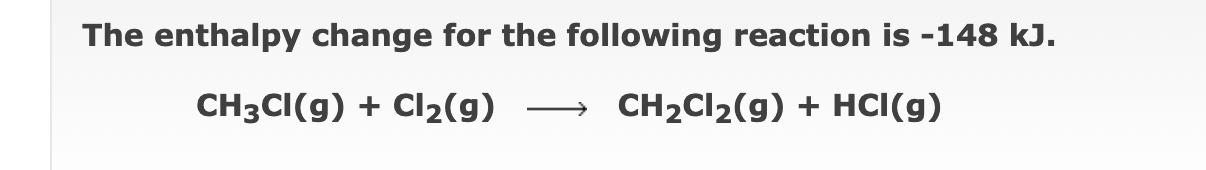
The enthalpy change for the following reaction is -148 kJ. CH3CI(g) + Cl(g) CHCl(g) + HCI(g)...
Fantastic news! We've Found the answer you've been seeking!
Question:





Transcribed Image Text:
The enthalpy change for the following reaction is -148 kJ. CH3CI(g) + Cl₂(g) CH₂Cl₂(g) + HCI(g) b Estimate the C-H bond energy in CH3CI(g), using tabulated bond energies (linked above) for the remaining bonds. C-H bond energy = kJ/mol The enthalpy change for the following reaction is -136 kJ. H₂(g) + O₂(g) H₂O₂(g) b Estimate the H-O bond energy in H₂O₂(g), using tabulated bond energies (linked above) for the remaining bonds. H-O bond energy = kJ/mol The enthalpy change for the following reaction is -566 kJ. 2CO(g) + O₂(g) →→2CO₂(g) b Estimate the O=0 bond energy in O₂(g), using tabulated bond energies (linked above) for the remaining bonds. O=O bond energy = kJ/mol The enthalpy change for the following reaction is -148 kJ. CH3CI(g) + Cl₂(g) CH₂Cl₂(g) + HCI(g) b Estimate the C-H bond energy in CH3CI(g), using tabulated bond energies (linked above) for the remaining bonds. C-H bond energy = kJ/mol The enthalpy change for the following reaction is -136 kJ. H₂(g) + O₂(g) H₂O₂(g) b Estimate the H-O bond energy in H₂O₂(g), using tabulated bond energies (linked above) for the remaining bonds. H-O bond energy = kJ/mol The enthalpy change for the following reaction is -566 kJ. 2CO(g) + O₂(g) →→2CO₂(g) b Estimate the O=0 bond energy in O₂(g), using tabulated bond energies (linked above) for the remaining bonds. O=O bond energy = kJ/mol
Expert Answer:
Answer rating: 100% (QA)
H 14 HCCL CCL HCCL HGH 1 H C1 Bond ralues this problem by taking t... View the full answer

Related Book For 

Chemistry The Central Science
ISBN: 978-0321696724
12th edition
Authors: Theodore Brown, Eugene LeMay, Bruce Bursten, Catherine Murphy, Patrick Woodward
Posted Date:
Students also viewed these physics questions
-
The standard enthalpy change for the following reaction is 436.4 kJ/mol: H2(g) - H(g) + H(g) Calculate the standard enthalpy of formation of atomic hydrogen (H)?
-
Calculate the enthalpy change for the following reaction: 3NO2(g) + H2O(l) 2HNO3(aq) + NO(g) Use standard enthalpies of formation.
-
The enthalpy change for each of the following reactions was calculated using bond energies. The bond energies of XO, YO, and ZO are all equal. XX + O=O XOOX; H = 275 kJ YY + O=O YOOY; H = +275 kJ...
-
Have leadership scholars, over the past 50 years, managed to produce a clear, cookie-cutter profile of the ideal leader through more than 1,000 studies
-
The new president of the Wernecke Company was stumped. Why had profits gone down? He had directed the sales department to push the product with the highest contribution margin , and the sales...
-
Linda has won the New Jersey megabucks lottery consisting of one million dollars. Suppose that she deposits the money in a savings account that pays an annual rate of 8% compounded continuously. How...
-
For each of the following organizational units, indicate whether the unit would most likely be classified as a cost center \((C)\), a revenue center \((R)\), a profit center \((P)\), or an investment...
-
A real-estate investor has the opportunity to purchase a small apartment complex. The apartment complex costs $4 million and is expected to generate net revenue (net after all operating and finance...
-
Week 3: Assignment1. The distribution of coffee consumption according to gender and neighborhood:Count the number of cases of low, moderate, and high coffeeconsumption for each gender. Count the...
-
From the tenth floor of her office building, Katherine Rally watches the swarms of New Yorkers fight their way through the streets infested with yellow cabs and the sidewalks littered with hot dog...
-
Danielle launches a ball off of a platform of height h with velocity vo at an angle 0 above the horizontal. A wall with a ball-sized hole a height H above the floor, with H > h, is a horizontal...
-
Find the volume V of the parallelepiped with the adjacent edges determined by the vectors: ~a =~i + 2~j + 3~ k,~b = ~i +~j + 2~ k, ~c = 2~i +~j + 4~ k, in the vector space V3.
-
Centrifugation is often used to thicken sludge solids. An incoming sludge with a solids concentration Co = 4% must be thickened to a solids concentration EC = 10% using a centrifuge. Unfortunately,...
-
Part A You must develop your own custom peripheral on FPGA and attach it to the embedded ARM processor on the Xilinx ZYNQ SOC via the AXI bus. The task is to implement an image filter (a convolution...
-
Shown here are annual financial data for a merchandising company and a manufacturing company. Music World Retail Wave-Board Manufacturing Beginning inventory Merchandise Finished goods Cost of...
-
Solve the system of two equations for y two equations are: A1=ax+by A2=cx+dy
-
Manufacturing plants create rolls of metal of a desired gauge (thickness) by feeding the metal through very large rollers. The thickness of the resulting metal depends on the gap between the working...
-
Following is the current balance sheet for a local partnership of doctors: The following questions represent independent situations: a. E is going to invest enough money in this partnership to...
-
A quantity of the blue solid on the left in Figure 13.7 is placed in an oven and heated for a time. It slowly turns from blue to the white of the solid on the right. What has occurred?
-
An ionic compound has a very negative in water. Would you expect it to be very soluble or nearly insoluble in water? Explain in terms of the enthalpy and entropy changes that accompany the process?
-
Calculate the concentration of an aqueous solution of NaOH that has a pH of 11.50.
-
White Corporation has 80,000 shares of \(\$ 5\) par value common stock outstanding. At year-end, the company declares a five percent stock dividend. The market price of the stock on the declaration...
-
Use the following data to prepare a statement of retained earnings for Barney Corporation. Total retained earnings originally reported at January 1... Cash dividends declared during the year. Net...
-
Bernard Corporation has the following shares outstanding: 8,000 shares of \(\$ 50\) par value, six percent preferred stock and 50,000 shares of \(\$ 1\) par value common stock. The company has \(\$...

Study smarter with the SolutionInn App


