The enzyme Rubisco constitutes up to 40% of the biomass in certain plant species, making it presumably
Question:
The enzyme Rubisco constitutes up to 40% of the biomass in certain plant species, making it presumably the most abundant enzyme in the world. It activates CO2 for the carboxylation of its substrate, ribulose-1,5-bisphosphate, thereby initiating the first step of carbon fixation in the Calvin cycle. The enzyme exhibits a moderate speed, so we estimate initial values for planning purposes with KM ⁓ 100 µM and kcat ⁓ 10 s-1.
a) For the reaction of Rubisco with its native substrate in the presence of CO2, calculate how far apart the time points of a kinetic experiment should be according to the initial velocity method to detect the product with an accuracy of 20 μM. Assume a (high) enzyme concentration of 100 μM and use the estimated values mentioned above, as well as the substrate concentrations in Table 5.1 (see below).
We conduct the experiment in a CO2-saturated solution and do not need to consider its concentration here.
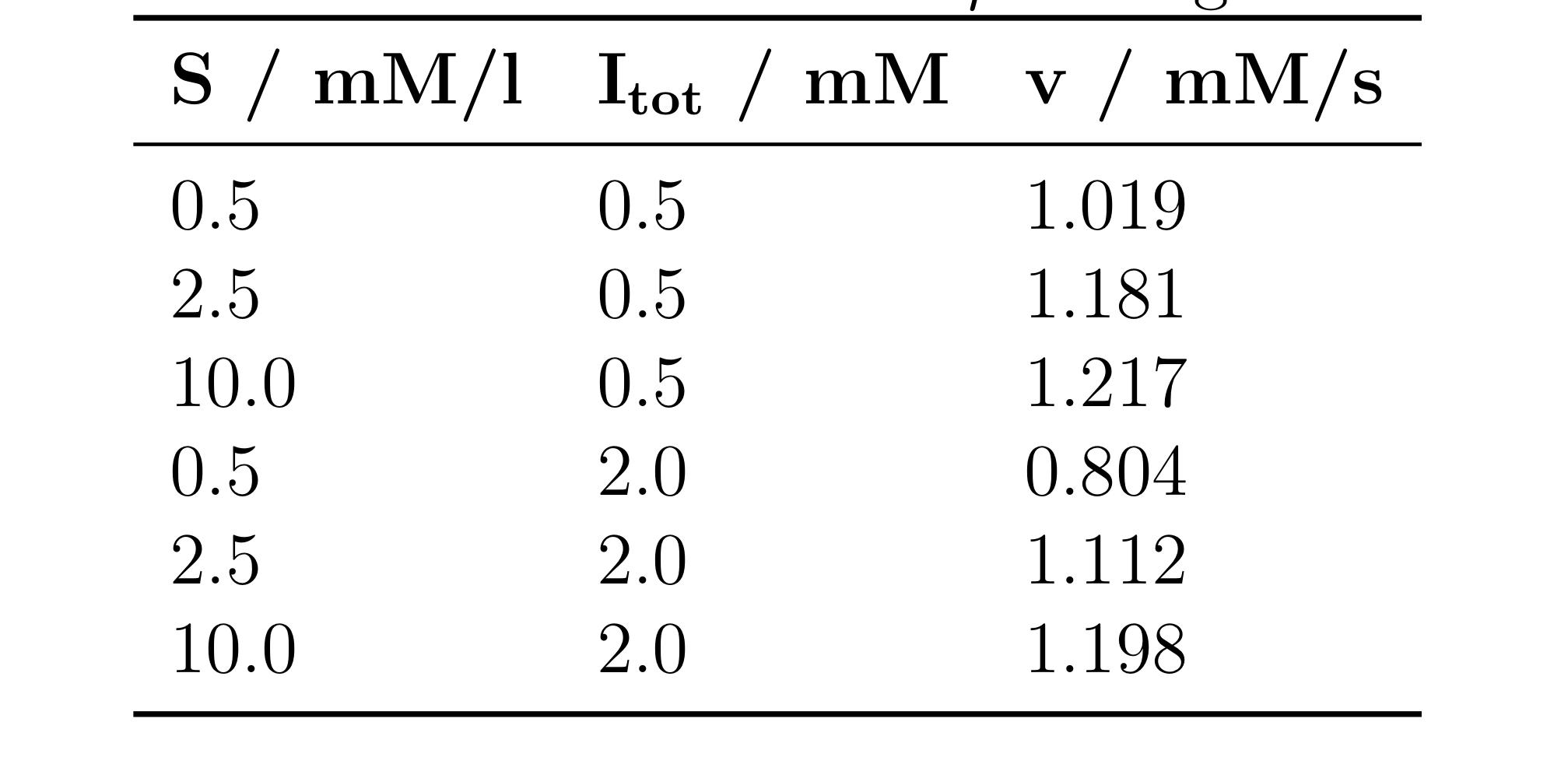
b) In the experiment, we conducted a trial with the competitive inhibitor D-fructose-1,6-bisphosphate and obtained the kinetic data in Table 5.1. Represent the data from Table 5.1 in an Eadie-Hofstee plot (i.e., a v vs. v/[S] diagram) and determine the enzyme's Michaelis constant as well as the inhibitor's dissociation constant, Kinh.
A sketch and calculation can easily substitute for a plot on the computer due to the limited number of data points.
c) What would change in your evaluation using an Eadie-Hofstee diagram in the case of an uncompetitive inhibitor? Describe, using two equations, firstly how the Eadie-Hofstee diagram looks in the case of an uncompetitive inhibitor, and secondly, how to interpret the slope and y-axis intercept in this diagram.

Managing Business Ethics Making Ethical Decisions
ISBN: 9781506388595
1st Edition
Authors: Alfred A. Marcus, Timothy J. Hargrave




