The equilibrium constant, K, for the following reaction is 1.8010 - at 698K. 2HI(g) H,(g) +...
Fantastic news! We've Found the answer you've been seeking!
Question:
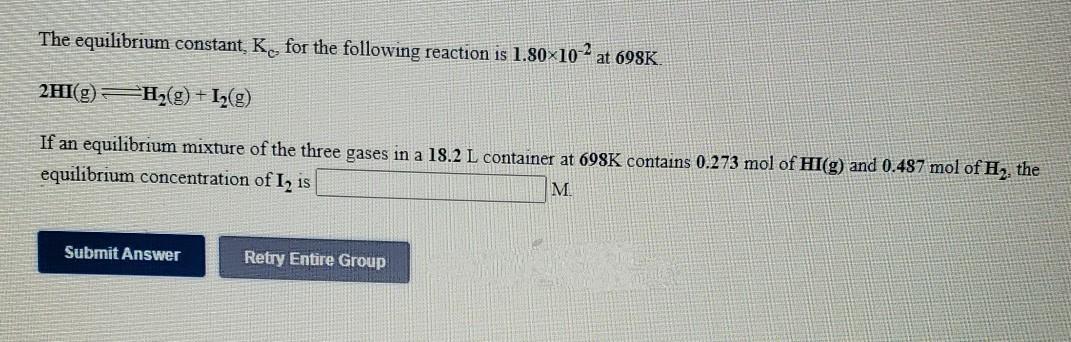
Transcribed Image Text:
The equilibrium constant, K, for the following reaction is 1.80×10 - at 698K. 2HI(g) H,(g) + I½(g) If an equilibrium mixture of the three gases in a 18.2 L container at 698K contains 0.273 mol of HI(g) and 0.487 mol of H, the equilibrium concentration of I, is M. Submit Answer Retry Entire Group The equilibrium constant, K, for the following reaction is 1.80×10 - at 698K. 2HI(g) H,(g) + I½(g) If an equilibrium mixture of the three gases in a 18.2 L container at 698K contains 0.273 mol of HI(g) and 0.487 mol of H, the equilibrium concentration of I, is M. Submit Answer Retry Entire Group
Expert Answer:
Related Book For 

Posted Date:
Students also viewed these chemistry questions
-
Calculate the equilibrium constant K for the following reaction at 25C from standard electrode potentials. Fe3 + (aq) + Cu(s) Fe2+(aq) + Cu2+(aq) The equation is not balanced.
-
Calculate the equilibrium constant K for the following reaction at 25C from standard electrode potentials. Sn4+(aq) + 2Hg(l Sn2+(aq) + Hg2 2+(aq) The equation is not balanced.
-
The equilibrium constant K for the reaction 2Cl(g) Cl2(g) was measured as a function of temperature (in kelvins). A graph of ln(K) versus 1/ T for this reaction gives a straight line with a slope of...
-
What type of business and industry is Fourevr Enterprises? What products or services do Fourevr Enterprises deliver?
-
What is the difference between lot size-based and volume-based quantity discounts?
-
Current Liability Entries and Adjustments Described below are certain transactions of Edward son Corporation. The company uses the periodic inventory system. 1. On February 2, the corporation...
-
Figure \(\mathrm{P} 27. 33\) shows the arrangement we looked at in Example 27. 2: a metal bar \(0.20 \mathrm{~m}\) long suspended from two springs, cach having a spring constant \(k=0.10 \mathrm{~N}...
-
Wood Incorporated factored $150,000 of accounts receivable with Engram Factors Inc. on a without-recourse basis. Engram assesses a 2% finance charge of the amount of accounts receivable and retains...
-
Discuss the following topic(s) in the forum and submit proof of your participation in the online discussions: 1) Professional certification and the practising accountants 2) Cost data and managerial...
-
This unadjusted trial balance is for Challenger Construction at the end of its fiscal year, September 30, 2023. The beginning balance of the owners capital account was $46,000 and the owner invested...
-
20. Set up the Filter Criteria area of the worksheet that defines the following criteria: - the Condition is equal to New and the Selling Price is greater than or equal to 25.99 - the Condition is...
-
ABC Company Ltd has 1.4 million shares common valued atTshs200 per share =Tshs280 million. Debt has face value ofTshs50 million and trades at 93% of face (Tshs46.5 million) in the market. Total...
-
it costs 1 5 0 x / 1 0 0 - x ( in millions of dollars ) to innoculate x percent of a population. How much of the population can we innoculate for $ 3 5 0 , 0 0 0 , 0 0 0 ?
-
Conduct a valuation of your company's stock Using the Graham Number Calculator. 2. What is used in this calculator to value the stock? What did the calculator suggest about the price of your stock?...
-
Patient presented at his regular doctor (in-network provider) because his ears hurt. The doctor diagnosed him with an ear infection and charged him a level 4 office visit for the covered charges...
-
Why following the FP Canada Professional Standards of Responsibility, FP Canada Annual Report, and IIROC (CRM, KYC) guidelines is important to Financial Planners, the profession and clients? If...
-
Jack goes to Big Sparkle to purchase supplies for $1920 on account Jack goes by Office De and purchases paper, ink, and coffee for office and pays $197 in cashiey Guardian Credit Union contracts with...
-
The population of Detroit, Michigan, decreased from 1,027,974 in 1990 to 688,701 in 2013 (Source: U.S. Census Bureau). Find the average rate of change in the population of Detroit, Michigan, over the...
-
Write structural formulas for the following compounds: (a) Trans-2-pentene. (b) 2-ethyl-1-butene, (c) 4-ethyl-trans-2-heptene. (d) 3-phenyl-butyne.
-
A 14-m by 10-m by 3.0-m basement had a high radon content. On the day the basement was sealed off from its surroundings so that no exchange of air could take place, the partial pressure of 222Rn was...
-
Use ionization energy and electron affinity values (see Table 8.3) to calculate the energychange (in kJ/mol) for the following reactions: (a) Li(g) + I(g) Li+ (g) + I - (g) (b) Na(g) + F(g) Na+ (g)...
-
Many states have lotteries that involve the random selection of digits 0, 1, 2, ,
-
Birth weights in the United States are normally distributed with a mean (in grams) of 3420 g and a standard deviation of 495 g. If you graph this normal distribution, the area to the right of 4000 g...
-
Is the distribution of those digits a normal distribution? Why or why not?

Study smarter with the SolutionInn App



