Question: The ether shown can be prepared using a Williamson ether synthesis; that is, by reacting an alkoxide ion with an alkyl halide. Draw the
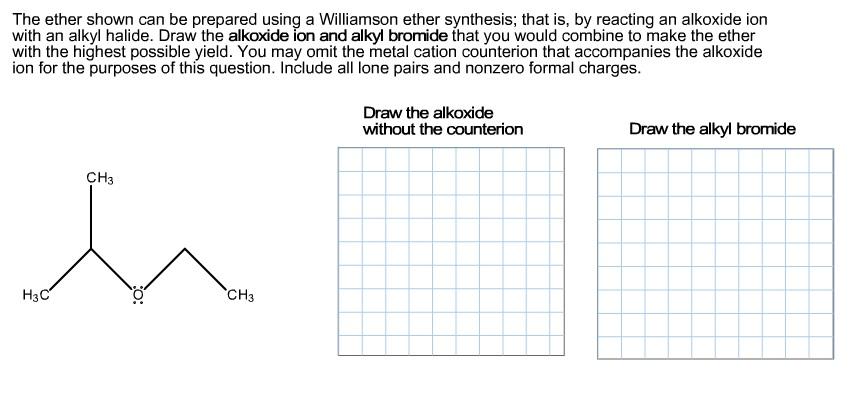
The ether shown can be prepared using a Williamson ether synthesis; that is, by reacting an alkoxide ion with an alkyl halide. Draw the alkoxide ion and alkyl bromide that you would combine to make the ether with the highest possible yield. You may omit the metal cation counterion that accompanies the alkoxide ion for the purposes of this question. Include all lone pairs and nonzero formal charges. Draw the alkoxide without the counterion Draw the alkyl bromide CH3 H3C CH3
Step by Step Solution
3.39 Rating (155 Votes )
There are 3 Steps involved in it

Get step-by-step solutions from verified subject matter experts


