The figure below provides steady-state data for a throttling valve in series with a heat exchanger....
Fantastic news! We've Found the answer you've been seeking!
Question:
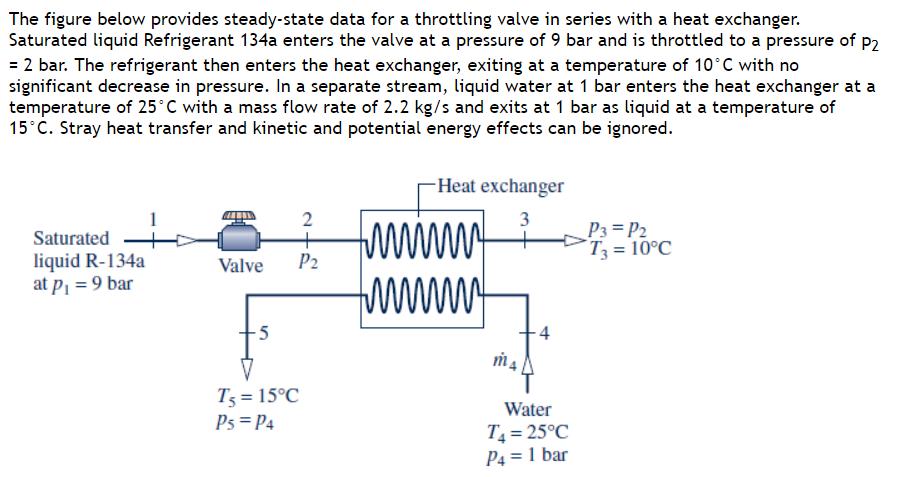

Transcribed Image Text:
The figure below provides steady-state data for a throttling valve in series with a heat exchanger. Saturated liquid Refrigerant 134a enters the valve at a pressure of 9 bar and is throttled to a pressure of P2 = 2 bar. The refrigerant then enters the heat exchanger, exiting at a temperature of 10°C with no significant decrease in pressure. In a separate stream, liquid water at 1 bar enters the heat exchanger at a temperature of 25°C with a mass flow rate of 2.2 kg/s and exits at 1 bar as liquid at a temperature of 15°C. Stray heat transfer and kinetic and potential energy effects can be ignored. Saturated liquid R-134a at P₁ = 9 bar 2 Valve P₂ 5 T5 = 15°C Ps= P4 -Heat exchanger 3 www www m4 4 Water T4=25°C P4 = 1 bar P3 = P2 T3 = 10°C Determine: (a) the temperature, in °C, of the refrigerant at the exit of the valve. T₂= °C (b) the mass flow rate of the refrigerant, in kg/s. m2 kg/s The figure below provides steady-state data for a throttling valve in series with a heat exchanger. Saturated liquid Refrigerant 134a enters the valve at a pressure of 9 bar and is throttled to a pressure of P2 = 2 bar. The refrigerant then enters the heat exchanger, exiting at a temperature of 10°C with no significant decrease in pressure. In a separate stream, liquid water at 1 bar enters the heat exchanger at a temperature of 25°C with a mass flow rate of 2.2 kg/s and exits at 1 bar as liquid at a temperature of 15°C. Stray heat transfer and kinetic and potential energy effects can be ignored. Saturated liquid R-134a at P₁ = 9 bar 2 Valve P₂ 5 T5 = 15°C Ps= P4 -Heat exchanger 3 www www m4 4 Water T4=25°C P4 = 1 bar P3 = P2 T3 = 10°C Determine: (a) the temperature, in °C, of the refrigerant at the exit of the valve. T₂= °C (b) the mass flow rate of the refrigerant, in kg/s. m2 kg/s
Expert Answer:
Answer rating: 100% (QA)
Solution a The temperature of the refrigerant at the exit of the valve T2 can ... View the full answer

Related Book For 

Thermodynamics An Engineering Approach
ISBN: 978-0073398174
8th edition
Authors: Yunus A. Cengel, Michael A. Boles
Posted Date:
Students also viewed these marketing questions
-
A two-stage compression refrigeration system operates between the pressure limits of 1 MPa and 0.12 MPa. The refrigerant, R-134a, leaves the condenser as saturated liquid and is throttled to a flash...
-
Three Zero Coupon Bonds (ZCB) are issued on the market. They each have face value of 100 and maturity, respectively, of 1 year, 3 years and 4 years. Assume the first bond is sold for 99.01, the...
-
Mel Morgan works as a cook for an Applebee's diner. His regular pay rate is $10 per hour, with time-and-a-half for hours in excess of 40 per week. Morgan's payroll deductions include withheld income...
-
A bank manager has developed a new system to reduce the time customers spend waiting to be served by tellers during peak business hours. Typical waiting times during peak business hours under the...
-
Discuss why AIDS is a reportable disease.
-
Calculate the child credit for the following taxpayers. Please show your work. a. Jeremy is a single (head of household) father with a dependent 8-year-old son and $79,600 of AGI:...
-
Lethe has borrowed $100 000 to purchase a drilling machine and promise to make 5 years of annual repayments at a fixed interest rate of 11.5% p.a. What is the annual repayment?
-
1. Calculate the internal growth rate and sustainable growth rate for S&S Air. What do these numbers mean? 2. S&S Air is planning for a growth rate of 12 percent next year. Calculate the EFN for the...
-
A design engineer proposes using a simple mechanism consisting of an inclined plane and a pulley arrangement to reduce the weight required to lift M, and he proposed a design as shown in Figure 2. H...
-
A freight train leaves city \(\mathrm{A}\) and heads for city \(\mathrm{B}\), which is \(44 \mathrm{~km}\) away, at \(12 \mathrm{~km} / \mathrm{hr}\). At the same time, a passenger train leaves city...
-
After driving due north at \(72 \mathrm{~km} / \mathrm{hr}\) for 2 hours, you stop for \(20 \mathrm{~min}\) and then head back south at \(108 \mathrm{~km} / \mathrm{hr}\) to reach your starting...
-
A box slides from rest down a \(2-\mathrm{m}\) long \(20^{\circ}\) incline. How long does it take the box to reach the bottom of the incline? \(\cdot\)
-
Compute the real interest rate using the exact formula and the approximation formula for each set of assumptions listed in (a) through (c). a. \(i=4 \% ; \quad \pi^{e}=2 \%\) b. \(i=15 \% ; \quad...
-
A grenade launcher shoots a grenade vertically upward at an initial speed of \(120 \mathrm{~m} / \mathrm{s}\). What are the vertical distances covered by it after (a) 6.0 s, (b) 12.0 s?
-
ONLINE MUSIC GIVES RISE TO DISTRIBUTION HEADACHES Who would be the marketing manager for a music label? You might be able to rub shoulders with the stars but ultimately, once the partying is over,...
-
Players A, B, and C toss a fair coin in order. The first to throw a head wins. What are their respective chances of winning?
-
A sealed electronic box is to be cooled by tap water flowing through the channels on two of its sides. It is specified that the temperature rise of the water not exceed 4C. The power dissipation of...
-
An adiabatic heat exchanger is used to heat cold water at 15C entering at a rate of 5 kg/s by hot water at 90C entering at a rate of 4 kg/s. If the exit temperature of hot water is 50C, the exit...
-
Reconsider Prob. 11-120. Using EES (or other) software, investigate the effect of the source temperature on the minimum rate of heat supply. Let the source temperature vary from 50 to 250oC. Plot the...
-
For the periodic processes shown below: a. Schedule the processes using an RMS policy. b. Schedule the processes using an EDF policy. In each case, compute the schedule for an interval equal to the...
-
For the periodic processes shown below: a. Schedule the processes using an RMS policy. b. Schedule the processes using an EDF policy. In each case, compute the schedule for an interval equal to the...
-
For the given periodic process execution times and periods (P1 has the highest priority), show how much CPU time of higher-priority processes will be required during one period of each of the...

Study smarter with the SolutionInn App


