The figure below shows the zinc blende unit cell (left) and actual ion packing (right). O...
Fantastic news! We've Found the answer you've been seeking!
Question:
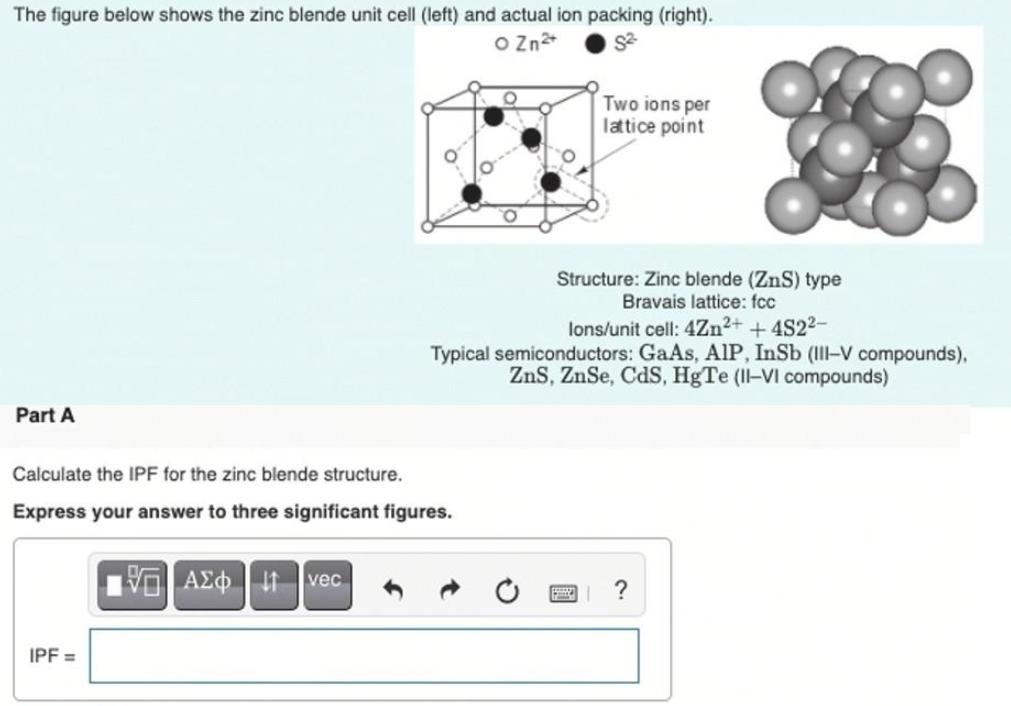
Transcribed Image Text:
The figure below shows the zinc blende unit cell (left) and actual ion packing (right). O Zn Two ions per latice point Structure: Zinc blende (ZnS) type Bravais lattice: fcc lons/unit cell: 4Zn2+ +4S22- Typical semiconductors: GaAs, AlP, InSb (III-V compounds), ZnS, ZnSe, CdS, HgTe (II-VI compounds) Part A Calculate the IPF for the zinc blende structure. Express your answer to three significant figures. vec IPF = The figure below shows the zinc blende unit cell (left) and actual ion packing (right). O Zn Two ions per latice point Structure: Zinc blende (ZnS) type Bravais lattice: fcc lons/unit cell: 4Zn2+ +4S22- Typical semiconductors: GaAs, AlP, InSb (III-V compounds), ZnS, ZnSe, CdS, HgTe (II-VI compounds) Part A Calculate the IPF for the zinc blende structure. Express your answer to three significant figures. vec IPF =
Expert Answer:
Answer rating: 100% (QA)
Answer Solution Ch zinc Blende tnit cell at Cakon Ahian E... View the full answer

Related Book For 

Posted Date:
Students also viewed these accounting questions
-
The figure below shows a set of human chromosomes. What is unusual about this person's genetic makeup? What health issues might this person suffer from? How does meiosis relate to this condition?
-
The figure below shows the market equilibrium (point B) at the intersection of demand and supply curves under perfect competition. Figure 11.5 D: Market demand curve S: Market supply curve Refer to...
-
The figure below shows an overhead view of an amusement park ride that travels at constant speed through five circular arcs of radii R0, 2R0, and 3R0. Rank the arcs according to the magnitude of the...
-
Please answer the following. Show your calculations for potential partial credit. Assume the expected return on the market is 14 percent and the risk-free rate is 4 percent. A.)What is the expected...
-
A horizontal analysis of a company's sales resulted in a $1.5 million increase, which equaled a percentage change of 22.8%. Required Interpret the dollar change and percentage change and identify...
-
Interscope sells the music of Lady Gaga, who promotes a unique public image and fashion style. Given her huge success, it is likely that by the end of the coming year, multiple performers will be...
-
Define what use case modeling is and is not.
-
Calculate the child credit for the following taxpayers. Please show your work. a. Jeremy is a single (head of household) father with a dependent 8-year-old son and $79,600 of AGI:...
-
Assume an investor buys a put option on XYZ stock with a strike price of $100 for $6 when the stock is trading for $97. What is the investor's maximum possible loss?
-
For the spring assemblages shown in Figures P2-8 through P2-16, determine the nodal displacements, the forces in each element, and the reactions. Use the direct stiffness method for all problems....
-
Kyle, a 85.0 kg football player, leaps straight up into the air (with no horizontal velocity) to catch a pass. He catches the 0.430 kg ball precisely at the peak of his jump, when he is 0.386 meters...
-
A snow-cone machine at an icecream shop costs \(\$ 15,000\). The machine is expected to generate profits of \(\$ 2,500\) each year of its 10-year useful life. At the end of the 10 years, the machine...
-
A small truck is purchased for \($17,000.\) It is expected to be of use to the company for 6 years, after which it will be sold for \($3,500.\) Determine the depreciation deduction and the resulting...
-
1. To develop an understanding of your ethical leadership style 2. To understand how your preferred ethical leadership style relates to other ethical leadership styles Directions 1. Please read the...
-
Consider the following Keynesian closed economy: Consumption \(\quad C=388+0.4(Y-T)-600 r\). Investment \(I=352-400 r\). Government purchases \(G=280\). Taxes \(\quad T=300\). Full-employment output...
-
What is the approximate magnitude of the electric field between the charge center and the ground? A. \(4 \times 10^{4} \mathrm{~V} / \mathrm{m}\) B. \(4 \times 10^{5} \mathrm{~V} / \mathrm{m}\) C....
-
Question 4 (1 point) Henry Co. has the following activity for their most recent year: Direct material used Direct Labor Manufacturing overhead Work in Process: Beginning Ending 40000 $39,000.00 $...
-
The polar coordinates of a point are given. Find the rectangular coordinates of the point. (-1, - /3)
-
Why do leakages reduce the money multiplier from its potential?
-
What role might foreign investment play in helping developing nations improve their growth rate and increase income levels?
-
In China, a small but increasing number of people are choosing to work as professional queuers, and exactly as it sounds, they are paid to wait in line for others. An NPR story in July 25, 2011,...
-
For the particular case of hard spheres, the pressure in the virial equation of state is determined by evaluating the pair correlation function at contact. Write the pair correlation function as...
-
Use a virial expansion approach to determine the first few nontrivial order contributions to the pair correlation function \(g(r)\) in \(d\) dimensions. Show that the pair correlation function is of...
-
Show that, in the case of a degenerate gas of fermions \(\left(T \ll T_{F} ight)\), the correlation function \(g(r)\), for \(r \gg \hbar / p_{F}\), reduces to the expression \[g(r)-1=-\frac{3(m k...

Study smarter with the SolutionInn App


