A line vortex of strength I m2/s is located at a distance of L m to...
Fantastic news! We've Found the answer you've been seeking!
Question:
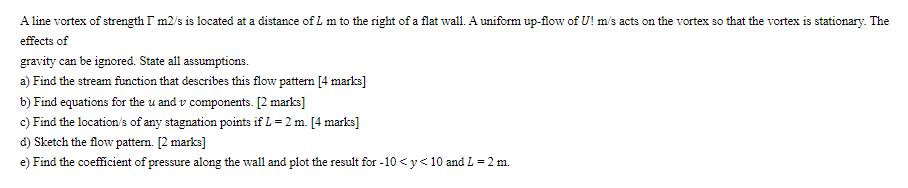
Transcribed Image Text:
A line vortex of strength I m2/s is located at a distance of L m to the right of a flat wall. A uniform up-flow of U! m/s acts on the vortex so that the vortex is stationary. The effects of gravity can be ignored. State all assumptions. a) Find the stream function that describes this flow pattern [4 marks] b) Find equations for the u and v components. [2 marks] c) Find the location/s of any stagnation points if L = 2 m. [4 marks] d) Sketch the flow pattern. [2 marks] e) Find the coefficient of pressure along the wall and plot the result for -10 <y<10 and L = 2 m. A line vortex of strength I m2/s is located at a distance of L m to the right of a flat wall. A uniform up-flow of U! m/s acts on the vortex so that the vortex is stationary. The effects of gravity can be ignored. State all assumptions. a) Find the stream function that describes this flow pattern [4 marks] b) Find equations for the u and v components. [2 marks] c) Find the location/s of any stagnation points if L = 2 m. [4 marks] d) Sketch the flow pattern. [2 marks] e) Find the coefficient of pressure along the wall and plot the result for -10 <y<10 and L = 2 m.
Expert Answer:

Related Book For 

Fundamental Accounting Principles
ISBN: 978-0078110870
20th Edition
Authors: John J. Wild, Ken W. Shaw, Barbara Chiappetta
Posted Date:
Students also viewed these accounting questions
-
The following unadjusted trial balance is for Crush Demolition Company as of the end of its April 30, 2011, fiscal year. The April 30, 2010, credit balance of the owners capital account was $36,900,...
-
The following unadjusted trial balance is for Ace Construction Co. as of the end of its 2013 fiscal year. The June 30, 2012, credit balance of the owners capital account was $53,660, and the owner...
-
The following unadjusted trial balance is for Braun Demolition Company as of the end of its April 30, 2011, fiscal year. Required 1. Prepare a 10-column work sheet for fiscal year 2011, starting with...
-
You are deciding between two mutually exclusive investment opportunities. Both require the same initial investment of $10 million. Investment A will generate $2 million per year (starting at the end...
-
On January 1, 2011, Lennon Corporation acquires 100% of Ono Inc. for $220,000 in cash. The condensed balance sheets of the two corporations immediately following the acquisition are as follows....
-
Your next assignment will be with the Cruise Director, Jacqueline Downs. She is concerned that passengers start their cruise vacation in a good frame of mind. Therefore, she is quite concerned about...
-
1. Identify an entrepreneur in your area you would like to interview. 2. Contact the person you have selected and make an appointment. Be sure to explain why you want the appointment and to give a...
-
(One Temporary Difference, Future Taxable Amounts, One Rate, No Beginning Deferred Taxes) Starfleet Corporation has one temporary difference at the end of 2010 that will reverse and cause taxable...
-
Corporate taxation in South Africa is too high and has negative implications for economic growth and our competitiveness with our main trading partners." Do you agree with this statement? Why (not)?...
-
The following is the statement of financial position of Rasheed Brothers Corporation (amounts in thousands). Instructions Evaluate the statement of financial position presented. State briefly the...
-
What stage in the PLC is the digital camera most likely in? a. growth b. maturity c. decline d. introductory
-
A company has a beta of 0.1, the T-bill rate is 6.68%, and the expected return on the market is 14.26%. What is its required rate of return? Do not round your intermediate calculations. Express as a...
-
a. Most currencies, such as fiat currencies (e.g. the U.S. Dollar), have a theoretically unlimited supply, while Bitcoin has a maximum fixed supply of only 18.7 million Bitcoin in total. Given the...
-
a.) What caused the recent increases in P&G products? b.) P&G announced 6% increase in sales revenue in the most recent quarter. Explain what we know is true of price elasticity given this increase...
-
1. Until roughly 2006, default rates on mortgages were fairly low due to ever increasing home prices. According to the videos, how did rising home prices make a cycle which then caused home prices to...
-
Given the coordinate matrix of x relative to a (nonstandard) basis B for R, find the coordinate matrix of x relative to the standard basis. B = {(1, 0, 0), (1, 1, 0), (0, 1, 1)}, []B =[4 0 2]T -01 E...
-
What effects are produced by an enzyme on the general reaction below? Select all that apply. S = P The formation of the transition state is promoted. The activation energy for the reaction is...
-
What do you think?
-
Refer to information in QS 24-14. Assume that actual sales are $265,000, actual variable costs for the year are $59,000, and actual fixed costs for the year are $73,400. Prepare a flexible budget...
-
Tytus Co. entered into the following transactions involving short-term liabilities in 2010 and 2011. 2010 Apr. 20 Purchased $38,500 of merchandise on credit from Frier, terms are 1y10, ny30. Tytus...
-
In December 2010, Monk Companys manager estimated next years total direct labor cost assuming 40 persons working an average of 1,500 hours each at an average wage rate of $50 per hour. The manager...
-
How many structural isomers are there for hydrocarbons that have the molecular formula C 4 H 10 ? (a) none (b) one (c) two (d) three
-
Which contains more hydrogen atomsa five-carbon saturated hydrocarbon molecule or a five-carbon unsaturated hydrocarbon molecule? (a) The unsaturated hydrocarbon has more hydrogen atoms. (b) The...
-
Explain why caprylic acid, CH 3 (CH 2 ) 6 COOH, dissolves in a 5, aqueous solution of sodium hydroxide but caprylaldehyde, CH 3 (CH 2 ) 6 CHO, does not dissolve. (a) With two oxygens, the caprylic...

Study smarter with the SolutionInn App


