The following data were taken from the records of Sweet Serendipity Co. before the accounts are...
Fantastic news! We've Found the answer you've been seeking!
Question:
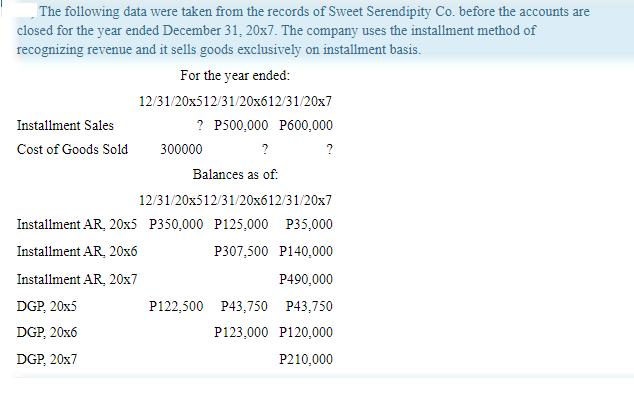
Transcribed Image Text:
The following data were taken from the records of Sweet Serendipity Co. before the accounts are closed for the year ended December 31, 20x7. The company uses the installment method of recognizing revenue and it sells goods exclusively on installment basis. Installment Sales Cost of Goods Sold Installment AR, 20x5 Installment AR, 20x6 Installment AR, 20x7 DGP, 20x5 DGP, 20x6 DGP, 20x7 For the year ended: 12/31/20x512/31/20x612/31/20x7 ? P500,000 P600,000 300000 ? Balances as of: 12/31/20x512/31/20x612/31/20x7 P350,000 P125,000 P35,000 P307,500 P140,000 P490,000 P122,500 P43,750 P43,750 P123,000 P120,000 P210,000 The following data were taken from the records of Sweet Serendipity Co. before the accounts are closed for the year ended December 31, 20x7. The company uses the installment method of recognizing revenue and it sells goods exclusively on installment basis. Installment Sales Cost of Goods Sold Installment AR, 20x5 Installment AR, 20x6 Installment AR, 20x7 DGP, 20x5 DGP, 20x6 DGP, 20x7 For the year ended: 12/31/20x512/31/20x612/31/20x7 ? P500,000 P600,000 300000 ? Balances as of: 12/31/20x512/31/20x612/31/20x7 P350,000 P125,000 P35,000 P307,500 P140,000 P490,000 P122,500 P43,750 P43,750 P123,000 P120,000 P210,000
Expert Answer:
Answer rating: 100% (QA)
ANSWER Installment Sales 20x5 P500000 20x6 P600000 20x7 P0 Cost of ... View the full answer

Related Book For 

Financial Accounting An Integrated Statements Approach
ISBN: 978-0324312119
2nd Edition
Authors: Jonathan E. Duchac, James M. Reeve, Carl S. Warren
Posted Date:
Students also viewed these accounting questions
-
The following data were taken from the records of Cougar Enterprises, a Canadian manufacturer that uses a normal job-order costing system: During December, the company worked on jobs numbered 70...
-
The following data were taken from the records of Surfs Up Corporation for the year ended July 31, 2006: Retained earnings and balance sheet data: Accounts payable ...........................$ 9,500...
-
The following data were taken from the records of Stellar Manufacturing Company for the fiscal year ended December 31, 2016: Instructions (a) Prepare a cost of goods manufactured schedule. (Assume...
-
TCP: the client sends only 1 message to the server hello from TCP Client and the server responds with the uppercase message. Update the program / make a simple chat program so that The client can...
-
About 70 percent of Internet users have actually purchased something online. Have you made an online purchase? If so, why do you think so many people who have access to the Internet are not also...
-
Each matrix is in Jordan form. State its characteristic polynomial and its minimal polynomial. (a) (b) (c) (d) (e) (f) (g) (h) 1 (e 2 00 1/ 020 210 003 031 310 0031 0300 3100 400 0 0 04 0 001_4/ 003...
-
Pilot Company had operating expense of \(\$ 40,000\). At the beginning of the year, Pilot owed \(\$ 8,000\) on accrued liabilities. At year-end, accrued liabilities were \(\$ 4,000\). How much cash...
-
Plastic Products, LLC, manufactures plastic beverage bottles. The division that manufactures water bottles for the North American market has two plants that operate 24 hours a day, 365 days a year....
-
Discuss the advanced computational techniques utilized in geotechnical engineering for modeling soil-structure interaction and predicting ground response under various loading conditions .
-
Which bond in Exhibit 1 most likely has the lowest effective convexity? A. AI bond B. BI bond C. CE bond Jules Bianchi is a bond analyst for Maneval Investments, Inc. Bianchi gathers data on three...
-
4. (20 points) For the network given in the figure below, fill out the datagram forwarding table for each node given below. The links are labeled with relative costs; your tables should forward each...
-
An auditor who discovers that a client's employees paid small bribes to municipal officials most likely would withdraw from the engagement if a. The payments violated the client's policies regarding...
-
Solve Chapter Problem 13.21, assuming the force is narrowband with a power spectral density given by \(S_{F}(\omega)=\frac{3 \times 10^{-3}}{2+5 \omega^{2}}\). Data From Chapter Problem 13.21: A SDOF...
-
Which of the following is the most important reason for an auditor to gain an understanding of an audit clients system of internal control over financial reporting? a. Understanding a clients system...
-
Which of the following statements best describes what is meant by an unqualified audit opinion? a. Issuance of an unqualified auditors report indicates that in the auditors opinion the clients...
-
Which of the following best places the events of the last several years in proper sequence? a. Sarbanes-Oxley Act, increased consulting services to audit clients, Enron and other scandals,...
-
Which of these is NOT a subject that may be found in a broker's post - license course? Advanced appraisal Advanced real estate and appraisal math Residential brokerage Real estate law
-
You continue to work in the corporate office for a nationwide convenience store franchise that operates nearly 10,000 stores. The per- store daily customer count (i.e., the mean number of customers...
-
The accounts receivable clerk for Vandalay Industries prepared the following partially completed aging-of-receivables schedule as of the end of business on November 30: The following accounts were...
-
A schedule of current assets and current liabilities for Lexmark International, Inc., a leading manufacturer of laser and inkjet printers, for three comparative years is as follows: In addition,...
-
On July 1, 2006, Cougar Corporation, a wholesaler of used robotic equipment, issued $7,500,000 of 10-year, 10% bonds when the market rate of interest was 13%. Interest on the bonds is payable...
-
Clausius-Clayperon equation is applicable to (a) Solid-vapour (b) Solid-liquid (c) Liquid-vapour (d) All of these.
-
The reaction \(A(\mathrm{l}) ightarrow R(\mathrm{~g})\) is allowed to reach equilibrium condition in an autoclave. At equilibrium there are two phases - one a pure liquid phase of \(A\) and the other...
-
The equilibrium constant for the reaction \(\mathrm{N}_{2}(\mathrm{~g})+3 \mathrm{H}_{2}(\mathrm{~g}) ightarrow 2 \mathrm{NH}_{3}\) is 0.1084 . Under the same conditions, the equilibrium constant for...

Study smarter with the SolutionInn App


