The following information has been extracted from the records of Due North Sales (DNS) Co.: January...
Fantastic news! We've Found the answer you've been seeking!
Question:
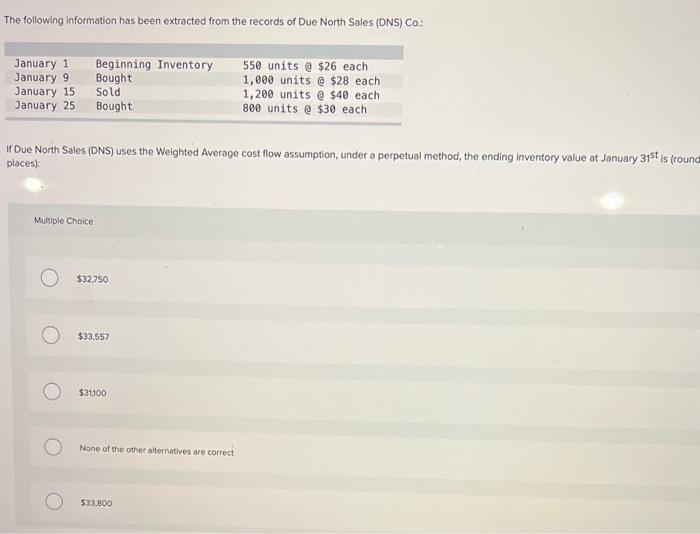
Transcribed Image Text:
The following information has been extracted from the records of Due North Sales (DNS) Co.: January 1 January 9 January 15 January 25 Beginning Inventory Bought Sold 550 units @ $26 each 1,000 units @ $28 each 1,200 units e $40 each 800 units @ $30 each Bought. If Due North Sales (DNS) uses the Weighted Average cost flow assumption, under a perpetual method, the ending inventory value at January 31st is (round places) Multiple Choice $32.750 $33,557 $31100 None of the other alternatives are correct 533.800 The following information has been extracted from the records of Due North Sales (DNS) Co.: January 1 January 9 January 15 January 25 Beginning Inventory Bought Sold 550 units @ $26 each 1,000 units @ $28 each 1,200 units e $40 each 800 units @ $30 each Bought. If Due North Sales (DNS) uses the Weighted Average cost flow assumption, under a perpetual method, the ending inventory value at January 31st is (round places) Multiple Choice $32.750 $33,557 $31100 None of the other alternatives are correct 533.800
Expert Answer:
Answer rating: 100% (QA)
Solution In case perpetual inventory records are maintained average rate of inventory is compute... View the full answer

Related Book For 

Accounting
ISBN: 978-1118608227
9th edition
Authors: Lew Edwards, John Medlin, Keryn Chalmers, Andreas Hellmann, Claire Beattie, Jodie Maxfield, John Hoggett
Posted Date:
Students also viewed these accounting questions
-
The following information has been extracted from the records of Target Company: Sales ....................................................................................$400,000 Purchases of direct...
-
The following information has been extracted from the records of Stevens Stationery about one of its popular products. Stevens Stationery uses the perpetual inventory system. Its annual reporting...
-
The following information has been extracted from the draft financial statements of TEX, a manufacturing entity: TEX - Income statement for the year ended 30 September 2003 September 2003...
-
Goddard Company has used the FIFO method of inventory valuation since it began operations in 2015. Goddard decided to change to the average cost method for determining inventory costs at the...
-
The epinephrine-mediated amplification cascade of Figure 24.14 has six steps, all of which are catalytic with one exception. This cascade leads to the activation of glycogen phosphorylase. This...
-
At the end of July, Tony took a complete inventory of his supplies and found the following: 5 dozen 14 screws at a cost of $8 a dozen 2 dozen 12 screws at a cost of $5 a dozen 2 cartons of computer...
-
For each of the following, calculate the standard error of the difference \(\left(s \mathrm{X}^{-} 1-\mathrm{X}^{-} ight.\) \(2)\). a. N = 35, s = 1.50, N = 35, $2 = 3.25 b. N = 4, s = 4.30, N = 4,...
-
Let us consider again the investment data from Hauck Financial Services used in Section 10.4 to illustrate the Markowitz portfolio model. The data follows, along with the return of the S&P 500 Index....
-
Flax Corp. uses the direct method to prepare its statement of cash flows. Flax's trial balances at December 31 for Year 6 and Year 5 show the following information: Account Cash Accounts Receivable...
-
Nielsen Media Research provides two measures of the television viewing audience: a television program rating, which is the percentage of households with televisions watching a program, and a...
-
A 8.0 nm thick cell membrane undergoes an action potential thatfollows the curve in the figure. Part A: Whatis the strength of the electric field inside the membrane justbefore the action potential?...
-
1 . The order is for 0 . 2 5 g of Ampicillin. The dose supplied is 1 2 5 mg Ampicillin / tablet . How many tablets are necessary?
-
Consider the following chemical reaction: CO (g) + 2H 2 (g) CH 3 OH(g) At equilibrium in a particular experiment, the concentrations of CO and H 2 were 0.15 M and 0.36M, respectively. What is the...
-
Draw 4-methylumbelliferone in its protonated and deprotonated state. Browse your files to upload or Drag and Drop Max attachments: 2 | Max Size: 20.00MB each
-
If an atom has an atomic number of 1 what is the element Olithium O argon O carbon hydrogen
-
4- Three conservation laws in nuclear reactions are conservation of charge conservation of baryon number conservation of lepton number. The reaction A. I. is proposed. Which conservation laws are...
-
2 Pat Hinkle earns $2,000.00 on a biweekly basis. Her overtime rate of pay per hour is a. $39.00 B b. $18.75 5 c. $37.50 Od. $25.00
-
Suppose the government bond described in problem 1 above is held for five years and then the savings institution acquiring the bond decides to sell it at a price of $940. Can you figure out the...
-
After reading the concepts of a good system of internal control covered in this chapter, the owner of a small business was concerned that his internal control was not good. He counted the daily cash...
-
Distinguish between the work performed by public accountants and the work performed by accountants in commerce and industry and in not-for-profit organisations.
-
Give an example of a transaction that results in: (a) An increase in one asset and an increase in a liability (b) A decrease in one asset but no change in the total assets (c) An increase in one...
-
Calculate the reduction ratio \(r_{k}=\frac{f\left(X_{k} ight)-f\left(X_{k}+s_{k} ight)}{m_{k}\left(X_{k} ight)-m_{k}\left(X_{k}+s_{k} ight)}\) for the function given in problem 1 .
-
Discuss the effect of initial trust region radius (ITRR) on the performance of TR algorithm.
-
Find the maximum of the function using Trust region method \(f=15 x_{1}+8 x_{1} x_{2}+5 x_{2}\) subject to \(x_{1}+x_{2} \leq 10\)

Study smarter with the SolutionInn App


